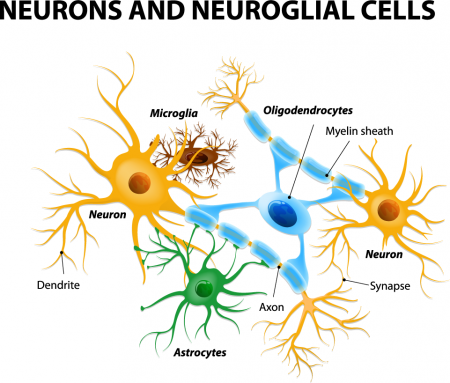
Astrocytes Can Turn Toxic
Astrocytes usually play a helpful role in the brain. These glial cells facilitate neural connections and prune unnecessary ones. However, new research details how infection or trauma can render astrocytes toxic, leading to brain disorders. In a 2017 article in the journal Nature, researcher Shane A. Liddelow and colleagues describe how resting astrocytes can become harmful reactive astrocytes.
The researchers determined that reactive astrocytes are found in brain tissues following brain injuries, or in neurological disorders including Alzheimer’s, Parkinson’s, Huntington’s, amyotrophic lateral sclerosis (ALS), and multiple sclerosis. Liddelow and colleagues also determined that microglia play a role in transforming resting astrocytes into a subtype of harmful reactive astrocytes, dubbed A1 astrocytes, and that the latter secrete a neuron-killing toxin.
A1 astrocytes lose their ability to promote neuronal survival and to create new synapses. The researchers showed that blocking the formation of A1 astrocytes prevented certain neuron deaths, and they hope this finding will lead to new treatments for brain injuries and neurological disorders.
Editor’s Note: It is possible that A1 astrocytes are also induced in patients with bipolar disorder, post-traumatic stress disorder (PTSD), and schizophrenia, as well as head traumas and neurological disorders.
Glia Cells Prune Over-Abundant Neurons
The brain contains neurons, which transmit electrical impulses, and glia, which protect and support neurons. New evidence suggests that some types of glia also play a role in pruning back overabundant neurons that are produced as the brain develops in utero.
Researcher Beth Stevens reports that astrocytes secrete a protein called transforming growth factor beta (TGF-beta). TGF-beta is a cytokine, or regulating protein, that activates brain microglia to initiate a complement cascade (C1 to C3), a series of chemical changes that destroy unnecessary neurons and synapses.
The various proteins involved in a complement cascade are numbered. This complement cascade starts with C1q and is continued by C4, C2, and C3, which initiate phagocytosis (or eating up) of the axon terminals of the underutilized neurons, sparing those that are active.
Inflammation and other changes in glia could cause either deficient or excess pruning of neurons, which has been thought to occur in neuropsychiatric disorders such as autism or schizophrenia.
Glial, Not Neuronal, Deficits May Cause Depression
The brain consists of 12 billion neurons and four times as many glial cells. Neurons conduct electrical activity, and it is thought that changes in neural activity and synaptic activity (where neurons meet) underlie most behaviors. It was once thought that glia were just fluff, but new research shows that they may play a role in depression.
There are three types of glia: astrocytes, oligodendrocytes, and mico-glia. Researcher Mounira Banasr had previously shown that neuronal lesions in the prefrontal cortex of mice did not produce depressive-like behaviors, but glial lesions did.
In a new study presented at a recent scientific meeting, Banasr reported that destroying astrocytes in the prefrontal cortex of mice induced depressive- and anxiety-like deficits. Using a virus that specifically targeted astrocytes, the researchers documented that the depressive behavior was specifically related to loss of astrocytes and not loss of other glial cell types, such as oligodendrocytes or micro-glia.
Editor’s Note: There is evidence of glial abnormalities in patients with mood disorders. Banasr’s research raises the possibility that glial deficits (rather than neuronal alterations) could be crucially involved in depression. In this study, the depressive- and anxiety-like behaviors persisted for 8 days following the astrocyte ablation, but by day 14 the animals had recovered, possibly with the production of a new supply of astrocytes. These data also raise the possibility that targeting the mechanisms of glial dysfunction could be a new avenue to pursue in the therapeutic approaches to depression.