Transcranial Direct Current Stimulation Improved OCD in Controlled Trial
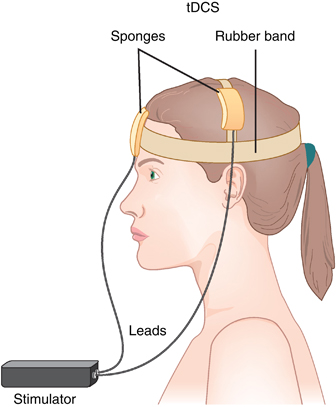
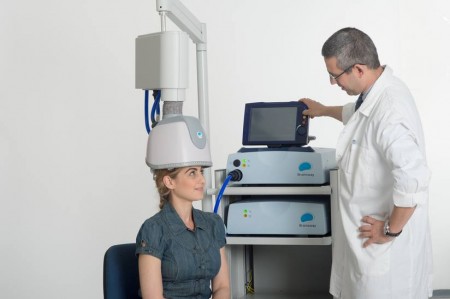
In an abstract of a paper that was to be presented at the 2020 meeting of the Society of Biological Psychiatry in May, researcher Roseli Gedanke Shavitt described a study of transcranial direct current stimulation (tDCS) in people with treatment-resistant obsessive-compulsive disorder (OCD). TDCS is a treatment in which electrodes applied to a patient’s scalp provide a constant low level of electricity that can modulate neuronal activity.
Shavitt and colleagues gave 30 minutes of either active or sham tDCS for 20 days to patients with treatment-resistant OCD. They positioned the cathode over the supplementary motor area of the brain, and the anode over the left deltoid. Those patients who received active tDCS achieved significantly greater reductions in OCD symptoms than did those in the sham group.
IVIG Produces Long-Term Results in PANDAS
PANDAS, or pediatric autoimmune neuropsychiatric disorder associated with streptococcal infection, is an autoimmune condition that produces psychiatric symptoms that appear suddenly following a case of strep throat in children. These symptoms can include obsessive-compulsive symptoms, tics, and behavioral dyscontrol and regression. Treatments are still experimental, but those that target the immune system are expected to be most successful at improving PANDAS.
In 2015, Miro Kovacevic and colleagues published a case series describing the use of intravenous immunoglobulin treatment (IVIG) in twelve children with PANDAS in the Journal of Child and Adolescent Psychopharmacology. One or in some cases two injections of IVIG brought about long-term remission in the children with PANDAS.
IVIG consists of a wide range of antibodies from multiple individuals delivered via injection. This increase in the quality or quantity of antibodies in the recipient is thought to suppress the production of antibodies that attack brain cells, causing PANDAS. The case series was based on patients at a large clinical practice that specializes in the treatment of PANDAS. The practice used a dosage of 1.5g/kg divided into two daily doses of 750 mg/kg, meant to match twice the volume of the patients’ own immunoglobulin G.
IVIG and other anti-inflammatory approaches are also effective in PANS, a more general variation on PANDAS in which psychiatric symptoms occur following an infection other than strep.
Deep TMS May Reduce OCD Symptoms
Obsessive compulsive disorder (OCD) occurs in about 2% of the population worldwide. Selective serotonin reuptake inhibitor (SSRI) antidepressants are the most commonly used treatment for OCD, but not all patients respond adequately to them.
At the 2015 meeting of the Transcranial Magnetic Stimulation Society, researcher Joseph Zohar presented evidence that deep transcranial magnetic stimulation (deep TMS) targeted over the medial prefrontal cortex may reduce OCD symptoms. In TMS treatment, an electromagnetic coil is placed against the patient’s head and magnetic pulses that can penetrate the scalp are converted into small electrical currents that stimulate neurons in the brain. In Zohar’s study, patients with OCD were randomized to receive deep TMS at frequencies of either 20 Hz or 1 Hz, or a sham procedure. The 20 Hz deep TMS resulted in a 28% reduction in OCD symptoms compared to the other two groups, indicating that the 20 Hz treatment had a large effect size.
In addition to the deep TMS procedures, all patients also received cognitive behavioral therapy, high doses of SSRIs, and relapse prevention training.
Editor’s Note: It is interesting that 20 Hz deep TMS, which activates the prefrontal cortex, was more effective than 1 Hz, which decreases activity there. Other attempts to treat OCD have focused on suppressing frontal-striatal-thalamic circuits, which are overactive in the disorder. Since the medial prefrontal cortex is an important area for the new learning required for the extinction of anxiety symptoms in a variety of disorders, increasing activity in this medial prefrontal target area with 20 Hz may activate that extinction process allowing new learning rather than nonspecifically suppressing hyperactive frontal-striatal-thalamic circuits as 1 Hz TMS would do.
PANS, an Inflammatory Disease with Psychiatric Symptoms in Kids
 Researcher Kiki Chang discussed pediatric acute onset neuropsychiatric syndrome (PANS), an inflammatory illness with psychiatric symptoms, at the 2014 meeting of the American Academy of Child and Adolescent Psychiatry. PANS is diagnosed when following an infection, a child who had previously been well has a sudden onset of obsessive-compulsive disorder (OCD), mood dysregulation, tics, food restriction behaviors, and a variety of other symptoms. A similar syndrome called PANDAS (for pediatric acute onset neuropsychiatic disease associated with streptococcal infections) was first identified in children recovering from strep throat. The children suddenly developed OCD behaviors and tics after a streptococcal infection.
Researcher Kiki Chang discussed pediatric acute onset neuropsychiatric syndrome (PANS), an inflammatory illness with psychiatric symptoms, at the 2014 meeting of the American Academy of Child and Adolescent Psychiatry. PANS is diagnosed when following an infection, a child who had previously been well has a sudden onset of obsessive-compulsive disorder (OCD), mood dysregulation, tics, food restriction behaviors, and a variety of other symptoms. A similar syndrome called PANDAS (for pediatric acute onset neuropsychiatic disease associated with streptococcal infections) was first identified in children recovering from strep throat. The children suddenly developed OCD behaviors and tics after a streptococcal infection.
However, PANS is associated with a variety of infections, including viruses and other infections that do not involve streptococcus bacteria. PANS syndrome is typified by acute onset of obsessive compulsive disorder and food restrictions as well as two or more of the following symptoms: anxiety, mood swings and depression, irritability and aggression, behavioral regression, decreases in school performance, sensory motor abnormalities, and somatic alterations such as decreased sleep and urinary incontinence, frequency, and/or urgency. Tics are not part of the formal diagnosis, but are present in about 50% of patients.
In Chang’s experience, the syndrome emerged 65% of the time in relationship to streptococcal infections, 13% with mycoplasma infections, 58% with viral infections, 39% in association with sinusitis, and 16% with otitis (inflammation of the ear). Increases in blood flow in the basal ganglia and increases in its volume likely occur due to antibodies that the immune system produces to fight infection, but which instead attack elements in the brain’s striatum, including tubulin, calcium calmodulin kinase II, lyso-GM-1, and dopamine D1 and D2 receptors.
Chang suggested that a diagnostic workup for PANS should include: a complete blood count and screening for red blood cell sedimentation rate, mycoplasma antibodies IgG and IgM, anti-nuclear antibodies (ANA), ferritin (a protein that stores iron in blood), celiac disease, and other laboratory measures that are commercially available in a panel produced by the company Moleculera Labs. A more detailed description of the PANS syndrome and its diagnosis and workup is available in the most recent 2014 issue of the Journal of the American Academy of Child and Adolescent Psychiatry.
In a related poster, Jennifer Frankovich, another researcher in Chang’s lab, reported that 62% of family members of children with PANS had a history of autoimmune disorders.
Topiramate Added to Regular Treatment Helps OCD symptoms in Bipolar Disorder
Until now, there has been little research about treating obsessive compulsive symptoms in people with bipolar disorder.
In a recent four-month double-blind placebo-controlled randomized clinical trial presented by Sharaian et al. at the 2014 meeting of the International Society for Bipolar Disorders, topiramate was more effective than placebo at reducing these symptoms in patients with bipolar disorder when added to their regular treatment. Nine of 17 study participants responded to topiramate (53%), while only two of 16 responded to placebo (12.5%).
Editor’s Note: These findings add to the list of comorbidities that topiramate may help with, even though it does not have any efficacy in the treatment of mania itself. Topiramate has helped with avoidance of cocaine and alcohol, bulimia and weight gain, anger attacks, and now obsessive compulsive disorder (OCD). Topiramate is also FDA-approved for migraine prevention in adolescents and adults.
Ketamine for OCD
At a recent scientific meeting, researcher Carolyn Rodriguez presented a randomized controlled crossover trial of ketamine in obsessive-compulsive disorder (OCD). In contrast to a previous negative study by Block and associates at the National Institute of Mental Health (NIMH), these investigators found that intravenous (IV) infusion of ketamine (0.5 mg/kg over 40 minutes) was associated with a larger reduction in obsessive-compulsive symptoms when compared with saline infusion. These effects were rapid in onset and persisted for approximately one week in 50% of the patients with OCD who had constant intrusive thoughts.
This dose of ketamine had previously been shown to induce rapid-onset improvement in depression and suicidal ideation in those with unipolar and bipolar depression. However, the improvement in obsessive-compulsive disorder symptoms appeared unrelated to any antidepressant effect because the individuals with OCD had minimal depressive symptoms at baseline.
The traditional pharmacological treatments for OCD are selective serotonin reuptake inhibitor (SSRI) antidepressants, which require high doses and weeks to months before the onset of full effect. In contrast, Rodriguez et al. found a 90% response rate to IV ketamine within 3 hours.
Ketamine is a blocker of the glutamate NMDA receptors, and these data suggest that targeting these receptors can induce rapid onset of positive effects in OCD. However, as is the case with the acute antidepressant response to ketamine in those with depression, the best ways to extend this therapeutic effect long-term remain to be determined.
Another blocker of NMDA receptors, the anti-Alzheimer’s drug memantine (Namenda), has been reported in open studies to show improvement in those with OCD as well. N-acetylcysteine, a substance found in health-food stores, likewise appears to re-regulate a hyper-responsive glutamatergic system in the nucleus accumbens by other mechanisms, and was also shown to have efficacy as an augmenting treatment in OCD in those who are inadequately responsive to SSRIs in a 2012 article by Afshaw et al.
Editor’s Note: Taken together, the data with ketamine, memantine, and N-acetylcysteine suggest that glutamate-based mechanisms are involved in OCD and may provide an alternative target for therapeutics in addition to serotonin.
Memantine May Be Useful in OCD, ADHD, and Autism
Memantine (Namenda) is an N-methyl-D-aspartate (NMDA) glutamate receptor antagonist that is FDA-approved for the treatment of Alzheimer’s dementia. Its use in other illnesses such as bipolar disorder and autism is currently being explored.
As we have written in previous issues of the BNN, A. Anand et al. reported in 2012 that in bipolar depression, memantine has an initial antidepressant augmentation effect when added to lamotrigine, an inhibitor of glutamate release. Koukopoulos et al. also reported in 2012 that in an open study, memantine had a large and sustained effect in previously treatment-resistant patients with bipolar disorder, producing an impressive 60-70% rate of excellent response at 6 months and again at 12 months of follow-up.
There is some evidence that memantine can be useful in obsessive-compulsive disorder (OCD). In a randomized double-blind placebo-controlled study of memantine combined with fluoxetine published by Ghaleiha et al. in 2012, patients with moderate to severe OCD taking memantine and fluoxetine were more likely to achieve remission after 8 weeks than patients taking placebo and fluoxetine.
Attention-deficit/hyperactivity disorder (ADHD) is another condition that memantine may be able to treat. Disturbances in NMDA receptor activity are thought to play a role in ADHD. Small, preliminary studies of memantine in ADHD have been promising.
New research has begun to explore memantine’s effects in autism. In one recent randomized, double-blind, placebo-controlled study published by Ghaleiha et al. in the International Journal of Neuropsychopharmacology, memantine produced improvement in children with autistic disorder when the drug was added to a treatment regimen that included risperidone, which blocks dopamine D2 receptors and is FDA-approved for the treatment of schizophrenia and mania, as well as autism.
However, at the 2012 meeting of the American Academy of Child and Adolescent Psychiatry, Robert Findling presented a poster on extended release memantine (Namenda) in children with autism, a study with negative results. This was a monotherapy study, unlike the above studies in which memantine was added to treatment with another drug. Findling found that extended release memantine (at doses of 3mg to 15mg per day) was well tolerated in children with autism, but the drug on its own was not significantly more effective than placebo in these preliminary studies.
Editor’s Note: Taken together, these data suggest an emerging role for memantine and possibly other drugs that work through NMDA receptor blockade in several disorders associated with repetitive behavior, like OCD and autism. The role of memantine augmentation in each of these syndromes deserves further exploration.