30 Minutes of TDCS Better Than 20 Minutes in Patients with Unipolar Depression
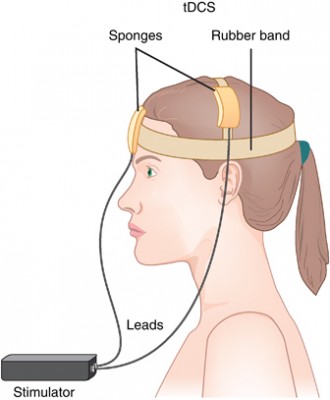 Transcranial direct current stimulation (tDCS) has successfully been used to treat depression. In this treatment, electrodes applied to the scalp provide a constant low level of electricity that can modulate neuron activity. In a 2017 article in the journal Progress in Neuro-Psychopharmacology & Biological Psychiatry, researcher Elena L. Pavlova and colleagues report that both 20- and 30-minute sessions of tDCS improved mild to moderate depression when combined with the selective-serotonin reuptake inhibitor (SSRI) antidepressant sertraline. However, the 30-minute sessions produced more improvement in depression.
Transcranial direct current stimulation (tDCS) has successfully been used to treat depression. In this treatment, electrodes applied to the scalp provide a constant low level of electricity that can modulate neuron activity. In a 2017 article in the journal Progress in Neuro-Psychopharmacology & Biological Psychiatry, researcher Elena L. Pavlova and colleagues report that both 20- and 30-minute sessions of tDCS improved mild to moderate depression when combined with the selective-serotonin reuptake inhibitor (SSRI) antidepressant sertraline. However, the 30-minute sessions produced more improvement in depression.
In the study, 69 right-handed patients (average age 37.6) received 50 mg of sertraline (Zoloft) per day and were randomized to one of three tDCS conditions: 10 daily 30-minute sessions, 10 daily 20-minute sessions, or 10 daily sham sessions with no tDCS treatment. The tDCS consisted of 0.5mA anodal current to the left dorsolateral prefrontal cortex.
Both 30-minute and 20-minute tDCS sessions produced greater benefit than the sham sessions. The 30-minute group showed significantly greater percentage improvement in depression scores than the 20-minute group, and included more participants who responded to treatment (89% compared to 68% of the 20-minute group and 50% of the sham group) and more whose depression remitted (70% compared to 27% of the 20-minute group and 35% of the sham group).
Creatine Supplements May Speed Up Response to Escitalopram, Improve Brain Connectivity
Antidepressants can take weeks to begin working, and researchers have been investigating ways to speed up this process. A 2012 study by In Kyoon Lyoo and colleagues in the American Journal of Psychiatry found that among 52 women taking the selective serotonin reuptake inhibitor (SSRI) antidepressant escitalopram (Lexapro) for unipolar depression, those who were prescribed an additional creatine supplement had earlier and greater decreases in depression symptoms than those who received a placebo in addition to the escitalopram.
The difference between the two groups was evident by the second week of treatment. At the end of the 8-week study, 52% of those who received creatine had achieved remission, compared to 26% of those in the placebo group.
Creatine, a supplement sometimes used by weightlifters, increases cellular energy. The women received 3g/day of creatine for the first week of the study, and 5g/day thereafter.
The same research group recently published more data from their creatine study. The new article by Sujung Yoon and colleagues in the journal Biological Psychiatry shows that following the creatine supplementation, the women in the creatine group had greater levels of N-acetylaspartate (a sign of healthy neurons) in their prefrontal cortex and also had greater levels of brain connectivity than women in the placebo group.
Thiamine (Vitamin B1) May Increase Effectiveness of Antidepressants
A new study suggests that the nutritional supplement vitamin B1, also known as thiamine, can improve symptoms of depression when taken with an antidepressant. Edith Holsboer-Trachsler and colleagues presented the research from their randomized, double-blind, placebo-controlled study at a recent scientific meeting. In a 12-week study, about 50 adults (averaging 35 years of age) with major depression were prescribed a selective-serotonin reuptake inhibitor (SSRI) antidepressant. In addition, half received thiamine supplements while the other half were given placebos. Starting at six weeks, those receiving thiamine with their antidepressant showed more improvement in their depressive symptoms than those receiving the antidepressant alone.
Thiamine is an essential nutrient for humans. It is found in foods such as yeast, pork, cereal grains, and certain vegetables. Thiamine deficiency has been linked to irritability and symptoms of depression, while thiamine supplementation can improve mood and reduce feelings of stress. No side effects were reported in the study.
Holsboer-Trachsler and colleagues hope that thiamine supplementation may help patients adhere to their antidepressant regimens by decreasing the time it takes until their moods begin to lift.
More Evidence that Statins Can Reduce Depression in People with Acute Coronary Syndrome
Many studies have linked depression and cardiovascular problems. The solutions may also be linked. A new study found that patients with depression and acute coronary syndrome saw their depression improve most when they took the selective-serotonin reuptake inhibitor (SSRI) antidepressant escitalopram and statins (used to lower cholesterol), while depression improved least among patients who took neither type of drug. Statin use was linked to improvement in depression after one year, while escitalopram was not. In a subset of the study, use of lipophilic statins in particular was linked to improvement in depression.
The study, published in 2015 by S.W. Kim and colleagues in the journal Translational Psychiatry, suggests that statins can improve depression regardless of antidepressant use, but combining statins with an SSRI may have an even more powerful effect on depression.
Genetic Variation Predicts Which Type of Antidepressant Will Be Effective
 In a six-month study of Caucasian patients, normal variations in the gene that is responsible for brain-derived neurotrophic factor (BDNF) predicted whether patients would respond better to a selective serotonin reuptake inhibitor (SSRI) antidepressant versus a serotonin and norepinephrine reuptake inhibitor (SNRI) or a tricycle antidepressant. There are several common variants of the BDNF gene, depending on which types of amino acids appear in its coding—valine or methionine. Patients with the most common version, two valines (or Val66Val), responded better to SSRIs. About two-thirds of the population has this version of the gene, which functions most efficiently. The remaining third have at least one methionine in the BDNF gene. Patients with a Met variation responded better to SNRIs and tricyclic antidepressants.
In a six-month study of Caucasian patients, normal variations in the gene that is responsible for brain-derived neurotrophic factor (BDNF) predicted whether patients would respond better to a selective serotonin reuptake inhibitor (SSRI) antidepressant versus a serotonin and norepinephrine reuptake inhibitor (SNRI) or a tricycle antidepressant. There are several common variants of the BDNF gene, depending on which types of amino acids appear in its coding—valine or methionine. Patients with the most common version, two valines (or Val66Val), responded better to SSRIs. About two-thirds of the population has this version of the gene, which functions most efficiently. The remaining third have at least one methionine in the BDNF gene. Patients with a Met variation responded better to SNRIs and tricyclic antidepressants.
The study by R. Colle and colleagues was published in the Journal of Affective Disorders in 2015. Of the patients who were prescribed SSRIs, 68.1% of patients with the Val/Val version responded to the medication after three months, compared to 44% of the patients with a Met version. Of patients prescribed SNRIs or tricyclics, 60.9% of the Met patients reached remission by six months, compared to only 33.3% of the Val/Val patients.
Editor’s Note: In an earlier BNN we reported that according to research published by Gonzalo Laje and colleagues in the journal Biological Psychiatry in 2012, depressed patients with the better functioning Val66Val allele of BDNF respond best to ketamine, while those with the intermediate functioning Val66Met allele respond less well.
Acupuncture with Paroxetine Better Than Paroxetine Alone
In a six-week study published by S.S. Qu et al. in the Journal of Psychiatric Research in 2013, participants with depression who received manual or electrical acupuncture along with the selective serotonin reuptake inhibitor (SSRI) antidepressant paroxetine (Paxil) improved more than those participants taking paroxetine alone.
More patients taking paroxetine alone needed increased doses to deal with symptom aggravation.
Patients who had received electrical acupuncture continued to show improvement four weeks after the treatment ended.
Oral Scopolamine Promising for Depression
Intravenous scopolamine has shown promise as a rapid-acting antidepressant in studies by Carlos Zarate and colleagues at the National Institute of Mental Health (NIMH). Improvement on the drug can occur within 24 hours.
In a 6-week 2012 study, an oral preparation of scopolamine was more effective than placebo as an add-on medication to the selective serotonin reuptake intake (SSRI) antidepressant citalopram. Patients who received scopolamine and citalopram had higher rates of response and remission than those who received placebo and citalopram. The scopolamine group experienced more blurred vision and dizziness, which is to be expected from an anticholinergic drug, a drug that blocks the action of the neurotransmitter acetylcholine in the brain.
Biomarker Suggests Which Patients With Depression Respond to Cognitive Behavior Therapy Versus SSRI
Not every treatment for mood disorders works for every patient, and for the 60% of depressed patients whose first treatment is ineffective, this wrong guess can translate into months of suffering, wasted money, lost productivity, and risk of suicide. An important trend in treatment research is the search for biomarkers, that is, biological indicators that can predict which patients might be likely (or unlikely) to respond to a particular treatment. A 2013 study by McGrath et al. in the journal JAMA Psychiatry suggests that brain glucose metabolism is one such biomarker.
Patients with untreated major depressive disorder had their brain glucose metabolism measured and then were randomized to receive 12 weeks of treatment either with the SSRI antidepressant escitalopram oxalate (trade name Lexapro) or with cognitive behavior therapy. Low glucose metabolism in a part of the brain called the anterior insula (compared to the rest of the brain) predicted that patients would reach remission on cognitive behavior therapy and respond poorly to escitalopram, while high metabolism in the same area predicted the opposite, that patients would reach remission while taking escitalopram and respond poorly to cognitive behavior therapy.
Researchers will want to test this finding with patients over the long term, but the data from this study suggest that anterior insula glucose metabolism may be a successful biomarker that can guide initial treatment selection for patients with depression.
Transcranial Direct Current Stimulation Plus Zoloft Has Better Antidepressant Effects Than Either Treatment Alone
 Transcranial direct current stimulation (tDCS), in which a barely perceptible level of electrical current is applied directly from one side of a patient’s scalp to the other, is a promising treatment for patients with tought-to-treat depression. A 2013 study by Brunoni et al. in JAMA Psychiatry examined whether combined treatment using tDCS and the selective-serotonin reuptake inhibitor (SSRI) antidepressant sertraline (Zoloft) would be a safe and effective treatment for unipolar depression. The combination was better than either treatment alone and better than placebo.
Transcranial direct current stimulation (tDCS), in which a barely perceptible level of electrical current is applied directly from one side of a patient’s scalp to the other, is a promising treatment for patients with tought-to-treat depression. A 2013 study by Brunoni et al. in JAMA Psychiatry examined whether combined treatment using tDCS and the selective-serotonin reuptake inhibitor (SSRI) antidepressant sertraline (Zoloft) would be a safe and effective treatment for unipolar depression. The combination was better than either treatment alone and better than placebo.
The six-week study used what is called a 2×2 factorial design, in which 120 patients with unipolar depression received either 50 mg/day of sertraline or placebo and also received either real tDCS or a sham procedure. The tDCS was administered in twelve 30-minute sessions, one per day Monday through Friday during the first two weeks, followed by one every other week. TDCS consists of an anodal (positive) and cathodal (negative) current placed at particular positions on the head. This study used 2 microamps of anodal left/cathodal right prefrontal stimulation for the tDCS treatment.
While the combination of sertraline and tDCS was significantly better than all three other treatment options (sertraline plus sham procedure, placebo plus tDCS, and placebo plus sham procedure), sertraline by itself and tDCS by itself resulted in similar efficacies. However, TDCS by itself was also significantly better than placebo, while sertraline by itself was not.
Side effects among the different treatment options were similar, except those who received tDCS had more scalp redness. There were seven instances of patients developing mania or hypomania during the study, five of which occurred in the combined tDCS and sertraline treatment group, higher than the 1–2% rate that would be expected in a study of unipolar depression.
Inflammatory Markers May Predict Antidepressant Response
There appears to be a link between inflammation and depression. In the journal Neuropsychopharmacology, Cattanes et al. reported in 2013 that compared to controls, depressed patients had significantly higher baseline levels of inflammatory cytokines, less glucocorticoid receptor function, less neuroplasticity, and fewer neuroprotective factors. Certain variables predicted response to treatment, others were seen only in responders, and still others changed in everyone with antidepressant treatment.
Higher baseline levels of inflammatory markers interleukin IB, macrophage inhibitory factor (MIF), and tumor necrosis factor TNF? were each associated with nonresponse to antidepressant treatment, and the three combined accounted for 50% of the variance in response—that is, they were the major predictor of whether a patient responded to treatment.
Levels of other factors changed in only those patients who responded well to antidepressants. The biggest changes were the normalization in levels of the neurotrophic factors BDNF and VEGF.
Several other markers normalized with antidepressant treatment regardless of whether the patients responded to treatment, and these included decreases in cytokines interleukin-IB and MIF and improved glucocorticoid receptor function.
The three different kinds of findings about these biomarkers were observed regardless of what type of antidepressant was used—SSRI versus tricyclic nortriptyline (which blocks norepinephrine reuptake).
Editor’s Note: This study replicates other studies in depression where signs of inflammation have been observed, including increases in inflammatory cytokines, decreases in glucocorticoid receptor function (needed to suppress high levels of the stress hormone cortisol) and lower levels of neuroplasticity and neuroprotection markers. This, however, is one of the first studies to show that levels of these markers at baseline may predict response to antidepressant treatment.
Also novel are the findings that while some high interleukin levels at baseline predicted antidepressant non-response, other ones normalized only in responders, and still others changed with treatment independent of whether the patients’ depression improved. These exciting findings require replication, but suggest the future possibility of personalized medicine, that is, choosing medications based on an individual biochemical marker profile. Eventually direct use of anti-inflammatory agents may be necessary in those with the highest levels of cytokines (predicting non-response to conventional antidepressant treatment). The same types of studies are needed in bipolar depression to determine the relationship between these inflammatory markers and treatment response.








