Poll Finds 87% of US Adults Think Children Need More Mental Health Support
In a poll of 2,014 adults in the US, 87% believed that children need more mental health support. For over a decade, the editors of the BNN have been emphasizing the great need for earlier recognition and treatment of children with bipolar and related mood and behavioral disorders. With so many Americans in agreement, now could be the time to direct public health and research funds to the better support of children’s mental health. Sadly this does not yet seem to be the case, but this is an election year. We need stand-up leaders in Congress to marshal the resources to increase the well-being of current and future generations.
Support Our Work by Donating!
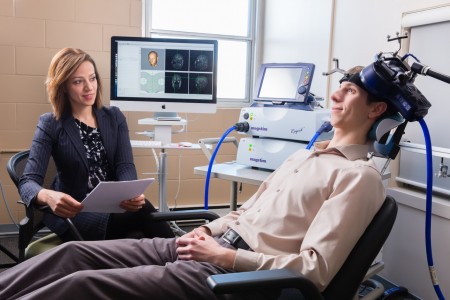
US Lags Behind Canada in Access to RTMS
At the 2015 meeting of the Transcranial Magnetic Stimulation Society, Linda Carpenter, an American researcher who specializes in repeated transcranial magnetic stimulation (rTMS), a method of treating depression by using a magnetic coil placed near the scalp to stimulate neurons, compared notes with Jeff Daskalakis, a Canadian researcher who also studies rTMS.
Carpenter described the limited approval rTMS enjoys in the US. RTMS has been approved by the Federal Drug Administration for the treatment of unipolar depression under very limited parameters (only at a frequency of 10Hz). RTMS has limited availability in the US, and many healthcare companies do not cover it. Providers face scrutiny of study recruitment practices and recordkeeping by insurers and the Joint Commission (formerly the Joint Commission of Accreditation of Healthcare Organizations), which assesses healthcare quality.
In contrast, Daskalakis and his Canadian colleagues can and do use rTMS to treat a broader range of illnesses including bipolar disorder. In Canada rTMS is used to treat unipolar depression, schizophrenia, post-traumatic stress disorder (PTSD), and obsessive-compulsive disorder (OCD), and clinicians can adjust the parameters to treat adolescents and the elderly.
The situation in the US is unfair. Because rTMS has not been approved for the treatment of bipolar disorder, Carpenter and other clinicians in the US are unable to treat bipolar depression even though a wide range of experts and published studies report that rTMS is as effective (or possibly even more so) for patients with bipolar depression than for those with unipolar depression.
Few treatments are available for bipolar depression. The discrepancy is even sadder when one considers that there are already more than 20 FDA-approved antidepressants that can be used to treat unipolar depression, but only three approved medications for bipolar depression. Bipolar depression is an orphan illness, which lacks a powerful voice advocating for more treatment research about optimal therapeutic strategies. Read more
The NIMH May Move Away from Using DSM-5
 The psychiatric community has been preparing for the 2013 release of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) by the American Psychiatric Association for years. Each new edition of the manual reflects changing conceptions of illnesses and their diagnosis and treatment.
The psychiatric community has been preparing for the 2013 release of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) by the American Psychiatric Association for years. Each new edition of the manual reflects changing conceptions of illnesses and their diagnosis and treatment.
Tom Insel, the director of the National Institute of Mental Health (NIMH), the largest research organization in the world devoted to the understanding and treatment of mental disorders, caused a stir this past spring when he wrote in a blog post that the DSM-5 lacks validity and that patients deserve better.
The DSM-5 guidelines for diagnosis of mental disorders rely on descriptions of clusters of symptoms, and Insel suggested that new methods of diagnosis that rely on laboratory measure should be developed. The NIMH is launching a project called Research Domain Criteria (RDoC) to incorporate genetics, imaging, and other data in a new classification system for illnesses, and is re-orienting their funding toward projects that “look across current categories,” for example by including all patients in a mood disorder clinic rather than only those who meet DSM-5 criteria for major depressive disorder.
A major concern about this change at the NIMH, which funds much of the research that leads to Federal Drug Administration-approved treatments, is that it will diminish funding for treatment studies in specific diagnostic categories where research is already sparse, such as childhood onset bipolar illness. This may leave many children and adults without a sound evidence base upon which their doctors can base treatment decisions.
Under the NIMH’s new rubric, clinical treatment studies to collect comparative data and evidence-based treatment research would likely lose out to studies focused on the broad collection and identification of biomarkers and the pursuit of new treatment targets. Answering an important clinical question such as whether symptoms of childhood onset bipolar disorder respond to the same medications as oppositional defiant disorder (ODD) or disruptive mood dysregulation disorder (DMDD) might not be a high priority for study.
David Kupfer, chair of the APA’s DSM-5 Task Force, responded to Insel’s statement saying that while it would be great to identify biomarkers and genetic indicators for mental illnesses, “this promise, which we have anticipated since the 1970s, remains disappointingly distant.” Insel acknowledged in his statement that this is only the beginning of development of research domain criteria, and that “for the present” the DSM will continue to be used.
Treatments Studies for Childhood Onset Bipolar Illness Are Inadequately Funded
It was remarkable that at the Pediatric Bipolar Conference hosted by Massachusetts General Hospital (MGH) and the Ryan Licht Sang Bipolar Foundation this past March in Cambridge, Massachusetts, none of the plenary talks, although they were excellent and given by leaders in the field of child psychiatry, dealt directly with the topic of the conference–childhood-onset bipolar disorder. There were also no reports of systematic placebo-controlled clinical trials evaluating treatment approaches in any of the subsequent presentations or posters. A number of open and uncontrolled studies examined new treatment possibilities.
It is notable that the National Institute of Mental Health (NIMH) no longer sponsors this conference, as it did for many years. Moreover, STEP-BD, an NIMH-sponsored research program on the course and treatment of adult-onset bipolar disorder, is now defunct, and the head of STEP-BD and one of the most productive researchers in bipolar illness, Andrew Nierenberg from the MGH, has been forced to search for other funding opportunities.
These developments highlight the ongoing deficient funding and study of both childhood-onset and adult-onset bipolar disorder despite the enormous public health impact, extraordinary morbidity, and early mortality from suicide and medical illnesses like cardiovascular disease that are associated with these disorders.