Simvastatin Looks Promising in Treatment of Negative Symptoms of Schizophrenia
The statin drug simvastatin (Zocor) enhances the effects of risperidone on negative symptoms of schizophrenia, according to a 2017 article by Soode Tajik-Esmaeeli and colleagues in the journal International Clinical Psychopharmacology.
In the 8-week study, 40 mg/day of simvastatin enhanced the effects of 4–6 mg/day of the antipsychotic risperidone on negative symptoms of schizophrenia, such as apathy and withdrawal, but not positive symptoms such as hallucinations or delusions.
Other statins, lovastatin and pravastatin, have not had a similar effect, possibly because they do not cross the blood-brain barrier as easily as simvastatin does.
Simvastatin has other benefits as well. Like all statins it decreases lipid levels, reducing cardiovascular disease. People with schizophrenia and bipolar disorder are at especially high risk for cardiovascular disease.
Simvastatin also decreases inflammation (lowering IL-1 alpha and TNF-beta levels) and may be neuroprotective, as it increases brain-derived neurotrophic factor (BDNF), a protein that protects neurons and is important for learning and memory. Inflammation is increasingly implicated in schizophrenia and bipolar disorder.
There is also some evidence that statins can prevent depressions over long-term follow-up. Studies in women without depression and men who had recently had heart attacks both showed that those taking statins had a lower rate of future depression than those not taking statins.
Editor’s Note: These findings suggest a potential 5-fold benefit to simvastatin: 1) It reduces negative symptoms in schizophrenia. 2) It reduces inflammation. 3) It increases BDNF. 4) It decreases cardiovascular disease risk by lowering lipid levels. 5) It may prevent future depressions.
Other approaches to augmenting schizophrenia treatment include nutritional supplements vitamin D3 and folate. Patients with psychosis often have vitamin D deficits. Folate supplements can reduce homocysteine, which has been linked to cognitive deficits in schizophrenia.
More Evidence N-Acetylcysteine Added to Risperidone Improves Irritability in Autism
We reported in 2014 that researchers Ahmad Ghanizadeh and Ebrahim Moghimi-Sarani had found that the over-the-counter nutritional supplement n-acetylcysteine (NAC) added to the atypical antipsychotic risperidone reduced irritability in autism more than placebo added to risperidone.
A randomized, double-blind, placebo-controlled clinical trial published by M. Nikoo and colleagues in Clinical Neuropharmacology in 2015 replicated these results. Forty children with autism disorders aged 4–12 years were randomized to receive either risperidone plus NAC or risperidone plus placebo. Risperidone doses were between 1 and 2 mg/day, and NAC doses were 600 to 900 mg/day. By the end of the 10-week study, those children who received NAC had significantly greater reductions in irritability and hyperactivity/noncompliance than those who received placebo.
Editor’s Note: Three placebo-controlled studies have supported the efficacy of NAC in autism. One 2012 study, by A.Y. Hardan in Biological Psychiatry, evaluated monotherapy with oral NAC. In the other two, NAC was added to treatment with risperidone.
Effect Size of Autism Treatments
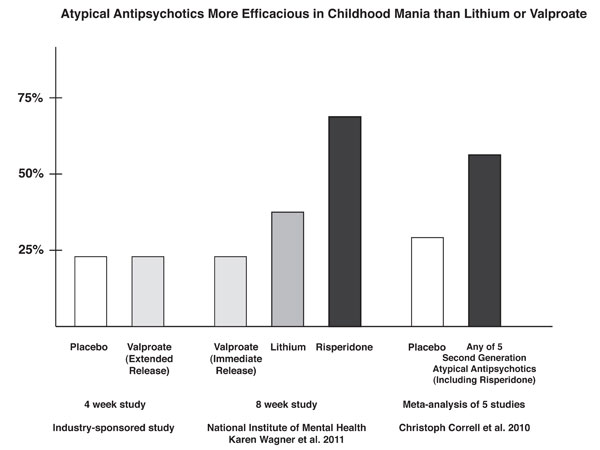
At the 2014 meeting of the American Academy of Child and Adolescent Psychiatry, Fung et al. presented a meta-analysis of treatments for autism that ranked them in terms of statistical effect size, ranging from 0.9 (large), to 0.5 to 0.8 (medium), to <0.4 (small). The only drug with a large effect size was risperidone, at 0.9. Most effect sizes were medium, including aripiprazole at 0.8 and N-acetylcysteine (NAC) at 0.7. Both clonidine and methylphenidate had effect sizes of 0.6, and tianeptine’s was 0.5.
Fung and colleagues noted that the first two on the list, the atypical antipsychotics risperidone and aripiprazole, often have problematic side effects (such as sedation, weight gain, and motor symptoms) that must be balanced against their effectiveness. In contrast, NAC is well tolerated with few side effects, and two placebo controlled studies showed that it was effective both alone and as an adjunctive treatment to the antipsychotic risperidone.
Combination of N-acetylcysteine and Risperidone Improves Irritability in Autistic Disorders More Than Placebo and Risperidone
In a 2013 study of 40 children and adolescents with autism spectrum disorders published by Ahmad Ghanizadeh and Ebrahim Moghimi-Sarani in the journal BMC Psychiatry, the combination of the over-the-counter nutritional supplement n-acetylcysteine (NAC) and the atypical antipsychotic risperidone alleviated irritability more than the combination of placebo and risperidone. Side effects were mild. The data extend 2012 observations by A.Y. Hardan et al. in which NAC improved irritability and stereotypy (repeated behavior) in autism more than placebo did.
The two studies taken together support the effectiveness of NAC prescribed either alone or in conjunction with an atypical antipsychotic for the treatment of autism.
Risperidone Trumps Valproate and Placebo for Treatment of Young Children with Mania
 At another symposium at the annual meeting of the American Academy of Child and Adolescent Psychiatry, Bob Kowatch of Ohio State University discussed a controlled trial of valproate, risperidone, and placebo in children 3 to 7 years of age (average age 5.5) with a diagnosis of bipolar I disorder and a Young Mania Rating Scale score (YMRS) greater than 20 at baseline. All of the children were severely ill with an average Clinical Global Assessment of Severity (CGAS) score of 44. Seventy-six percent had comorbid attention deficit hyperactivity disorder (ADHD) and 15% had an anxiety disorder. Valproate doses started at 10mg/kg and were increased after 4 days to achieve blood levels of 80 to 100µg/ml. The average dose of valproate was 300mg/day and the average blood level was 88 µg/ml. Risperidone was started at 0.25mg and increased as needed. The average dose of risperidone was 0.5mg per day.
At another symposium at the annual meeting of the American Academy of Child and Adolescent Psychiatry, Bob Kowatch of Ohio State University discussed a controlled trial of valproate, risperidone, and placebo in children 3 to 7 years of age (average age 5.5) with a diagnosis of bipolar I disorder and a Young Mania Rating Scale score (YMRS) greater than 20 at baseline. All of the children were severely ill with an average Clinical Global Assessment of Severity (CGAS) score of 44. Seventy-six percent had comorbid attention deficit hyperactivity disorder (ADHD) and 15% had an anxiety disorder. Valproate doses started at 10mg/kg and were increased after 4 days to achieve blood levels of 80 to 100µg/ml. The average dose of valproate was 300mg/day and the average blood level was 88 µg/ml. Risperidone was started at 0.25mg and increased as needed. The average dose of risperidone was 0.5mg per day.
On the main outcome measure of decrease in the YMRS score risperidone was substantially more effective than placebo, while valproate showed only marginal nonsignificant effects. However on the Clinical Global Impressions (CGI) scale for improvement in illness, risperidone showed 87% response, valproate 75% response, and placebo no response. In terms of 50% reduction in the YMRS score, this endpoint was achieved in 88% on risperidone, 50% valproate, and 15% on placebo.
Weight gain was mild on valproate and substantially more on risperidone. Risperidone was also associated with increases in insulin and prolactin.
The effect size (the size of the change the drug brought about in this study, which is calculated by dividing the mean difference between the experimental group and the control group by the standard deviation) for risperidone was extraordinarily large (3.58); very large for valproate (1.66), and moderate for placebo (0.56). The odds of getting well were 5 times greater than placebo for risperidone and 1.9 times greater than placebo for valproate.
Editors note: These data in very young children (aged 3 to 7) resemble other controlled data in the literature about the treatment of older children and adolescents, indicating a superiority of atypical antipsychotics over placebo and a greater magnitude of effect achieved with atypicals than with valproate. Based on these new data and the Federal Drug Administration (FDA) approval of several atypical antipsychotics for children with bipolar illness from ages 10 to 17, Dr. Kowatch recommended a new treatment algorithm for childhood onset bipolar disorder. Read more
News From The “TEAM” Study: Risperidone Superior To Valproate And Lithium In Childhood Mania
A symposium at the Annual Meeting of the American Association of Child and Adolescent Psychiatry discussed the Treatment of Early Age Mania (TEAM) study, which comprised 5 different sites in Pittsburgh, Washington DC, Baltimore, St. Louis, and Cleveland. This randomized partially blinded study compared risperidone, valproate, and lithium for the treatment of children with bipolar I mania.
Participants were all severely ill with a Clinical Global Assessment of Severity score (C-GAS) of less than 60 (the mean was 39, indicating that the children were substantially impaired). More than three quarters had psychosis (i.e. hallucinations or delusions) and 99% had dramatic mood shifts within a day (ultradian cycling). All the children had the cardinal symptom of elevated mood.
Among the 290 participants, there was a high incidence of Axis I comorbidities, with 98% of patients having a disruptive behavioral disorder, 77.3% an anxiety disorder, 31% some form of sleep disturbance, and 17% an elimination disorder, of which 15% had enuresis (bedwetting). Nightmares were present in 25.9% of the sample, sleepwalking in 7.2%, and night terrors in 4.8%.
For the purposes of the study, response was considered to have been achieved when a child received a rating of 1 (not ill) or 2 (minimally ill) on the Clinical Global Impressions scale modified for bipolar illness (CGI-BP).
The children (age 6 to 15 with a mean age of 11) were randomized to treatment for 8 weeks with lithium, valproate, or risperidone. Lithium treatment reached blood levels of 1.1 to 1.3mEq/L, valproate reached levels of 111 – 225µg/ML, and risperidone doses were up to 3mg per day. Children who were taking psychomotor stimulants for treatment of ADHD remained on the stimulants after randomization to one of the three drugs. While the treating physicians and clinicians were not blind, blind ratings were performed at week 8.
With a response rate of 68.5%, risperidone was superior to lithium (35.6%) and valproate (24%) based on CGI-BP scores. The mean dose of risperidone was 2.6mg +/- 1.2 per day. The mean blood level at week 8 for lithium was 1.1mEq/L and for valproate was 114µg/ML.
The number of children who improved sufficiently for their C-GAS scores to rise above 60 was also greater for risperidone at 48.3% compared to lithium at 26.7% and valproate at 17.0%. Read more
Valnoctamide Effective in Mania, Maybe Without Valproate’s Side Effects
 Valproate (Depakote), also known as divalproex sodium and valproic acid (VPA), is highly effective in the treatment of mania, seizures, and migraine. However, its use in pregnant mothers can cause birth defects and developmental delay. A closely related compound, valnoctamide, may not pose the same dangers, but its efficacy in mania has only recently been investigated.
Valproate (Depakote), also known as divalproex sodium and valproic acid (VPA), is highly effective in the treatment of mania, seizures, and migraine. However, its use in pregnant mothers can cause birth defects and developmental delay. A closely related compound, valnoctamide, may not pose the same dangers, but its efficacy in mania has only recently been investigated.
Yuly Bersudsky et al. reported at the 4th Biennial Conference of the International Society for Bipolar Disorders conference in Sao Paulo, Brazil in March that valnoctamide was more effective than placebo as an add-on to risperidone for the treatment of mania.
Since there is now evidence that valnoctamide does work in mania, it is plausible that some of the shared characteristics of valproate and valnoctamide, such as increasing brain GABA and blocking sodium channels, are responsible for both drugs’ antimanic effects. Read more
Faster, Better Response to Risperidone than Valproate in Adolescents with Bipolar Disorder
An article by Pavuluri et al. published in Bipolar Disorders in September reported that both divalproex sodium (valproate, or Depakote) and risperidone (Risperdol) were effective in youth with bipolar disorder, but improvements appeared more quickly with risperidone. Risperidone also produced higher response rates, higher remission rates, and fewer dropouts from side effects.
A presentation by the research group at an earlier conference suggested that it was particularly among those with comorbid disruptive behavioral disorders (DBD), which include attention deficit hyperactivity disorder (ADHD), oppositional defiant disorder (ODD), and conduct disorder (CD), that risperidone worked faster and produced greater early results than divalproex.
 In the study, 66 children with type I bipolar disorder and a mean age of 11 years were assessed. Treatment with risperidone was initiated at 0.5 mg/day and titrated to 2 mg, while divalproex was initiated at 60 micrograms/mL and titrated up to 120 micrograms.
In the study, 66 children with type I bipolar disorder and a mean age of 11 years were assessed. Treatment with risperidone was initiated at 0.5 mg/day and titrated to 2 mg, while divalproex was initiated at 60 micrograms/mL and titrated up to 120 micrograms.
Editor’s Note: The possibility that children with different comorbid disorders respond differently to different antimanic agents suggests that more studies are needed to determine which subgroups of patients are most responsive to typical treatments.
One Expert’s Personal Treatment Algorithm for Bipolar Disorder in Young Children
EDITOR’S NOTE: Dr. Gagin Joshi of Massachusetts General Hospital, who presented the work on carbamazepine and lamotrigine on page 1 provided us with his own general treatment algorithm for youngsters with bipolar disorder.

Omega-3 Fatty Acids (photo from ironmagazine.com)
Joshi typically starts with 0.5 to 2 gms of omega-3 fatty acids because of their benign side-effects profile, the many studies suggesting they are effective in adult mood disorders, and a recent article indicating that they were effective in preventing the conversion of prodromal schizophrenia into full-blown illness in a randomized double-blind controlled study in Australia.
After the omega-3 fatty acids, Joshi’s second choice is typically the atypical antipsychotic aripiprazole (Abilify) because of its lesser degree of weight gain compared to atypicals quetiapine (Seroquel) or risperidone (Risperidol). Risperidone can be a third option if aripiprazole is not effective or tolerated.