Three Experts’ Different Approaches to Treating PTSD in Veterans
 In the BNN we have previously described some experts’ preferred treatment algorithms for patients with treatment-resistant post-traumatic stress disorder (PTSD), which is often complicated by traumatic brain injury (TBI). In this article, we update and expand upon these expert views.
In the BNN we have previously described some experts’ preferred treatment algorithms for patients with treatment-resistant post-traumatic stress disorder (PTSD), which is often complicated by traumatic brain injury (TBI). In this article, we update and expand upon these expert views.
David Bakish has worked as Medical Director at the Ottawa Psychopharmacology Clinic and is a former professor of psychiatry at the University of Ottawa in Ottawa, Ontario. In addition, he works with the Canadian military seeing patients with PTSD, substance abuse, and traumatic brain injuries. He uses a symptom-driven approach to PTSD, including 6 to 7 targeted medications added in sequence.
Albert Sattin is a professor of psychiatry and biobehavioral sciences at UCLA, belongs to their Brain Research Institute, and is affiliated with both the Ronald Reagan UCLA Medical Center and the Veterans Affairs Greater Los Angeles Healthcare System. He prefers to treat PTSD with a three-part combination of the blood pressure–lowering drug prazosin, a selective serotonin reuptake inhibitor (SSRI) antidepressant, and the atypical antidepressant mirtazapine.
Murray Raskind pioneered placebo-controlled studies of prazosin for PTSD and served as director of the Veterans Affairs Puget Sound Health Care System Mental Health Service, in addition to serving in the Department of Psychiatry and Behavioral Sciences at the University of Washington School of Medicine. Raskind’s approach to PTSD includes prazosin, the tricyclic antidepressant amitriptyline, and if needed for sleep, the sedative zolpidem.
Only SSRIs are approved by the US Food and Drug Administration (FDA) for the treatment of PTSD, but these on their own are rarely sufficient to handle the insomnia and other symptoms that accompany PTSD. Exposure therapy, in which patients are gradually led to approach trauma-related memories, feelings, and situations they previously avoided, is the most recommended type of therapy, but it too is often insufficient to treat all the complexities of the illness. Read on for more on each doctor’s approach to treating PTSD. Read more
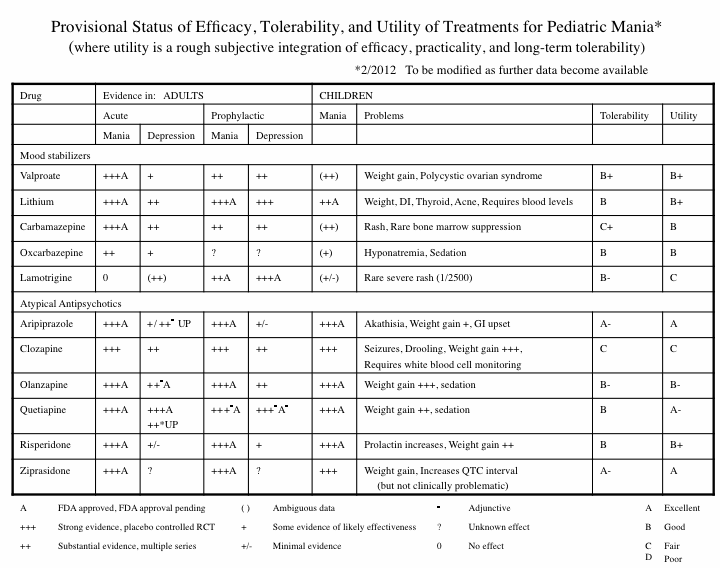
Efficacy, Tolerability, and Utility of Treatments for Pediatric Mania
The following table analyzes the information currently available about various treatment options (including treatments NOT yet FDA approved for the treatment of children):
Risperidone Trumps Valproate and Placebo for Treatment of Young Children with Mania
 At another symposium at the annual meeting of the American Academy of Child and Adolescent Psychiatry, Bob Kowatch of Ohio State University discussed a controlled trial of valproate, risperidone, and placebo in children 3 to 7 years of age (average age 5.5) with a diagnosis of bipolar I disorder and a Young Mania Rating Scale score (YMRS) greater than 20 at baseline. All of the children were severely ill with an average Clinical Global Assessment of Severity (CGAS) score of 44. Seventy-six percent had comorbid attention deficit hyperactivity disorder (ADHD) and 15% had an anxiety disorder. Valproate doses started at 10mg/kg and were increased after 4 days to achieve blood levels of 80 to 100µg/ml. The average dose of valproate was 300mg/day and the average blood level was 88 µg/ml. Risperidone was started at 0.25mg and increased as needed. The average dose of risperidone was 0.5mg per day.
At another symposium at the annual meeting of the American Academy of Child and Adolescent Psychiatry, Bob Kowatch of Ohio State University discussed a controlled trial of valproate, risperidone, and placebo in children 3 to 7 years of age (average age 5.5) with a diagnosis of bipolar I disorder and a Young Mania Rating Scale score (YMRS) greater than 20 at baseline. All of the children were severely ill with an average Clinical Global Assessment of Severity (CGAS) score of 44. Seventy-six percent had comorbid attention deficit hyperactivity disorder (ADHD) and 15% had an anxiety disorder. Valproate doses started at 10mg/kg and were increased after 4 days to achieve blood levels of 80 to 100µg/ml. The average dose of valproate was 300mg/day and the average blood level was 88 µg/ml. Risperidone was started at 0.25mg and increased as needed. The average dose of risperidone was 0.5mg per day.
On the main outcome measure of decrease in the YMRS score risperidone was substantially more effective than placebo, while valproate showed only marginal nonsignificant effects. However on the Clinical Global Impressions (CGI) scale for improvement in illness, risperidone showed 87% response, valproate 75% response, and placebo no response. In terms of 50% reduction in the YMRS score, this endpoint was achieved in 88% on risperidone, 50% valproate, and 15% on placebo.
Weight gain was mild on valproate and substantially more on risperidone. Risperidone was also associated with increases in insulin and prolactin.
The effect size (the size of the change the drug brought about in this study, which is calculated by dividing the mean difference between the experimental group and the control group by the standard deviation) for risperidone was extraordinarily large (3.58); very large for valproate (1.66), and moderate for placebo (0.56). The odds of getting well were 5 times greater than placebo for risperidone and 1.9 times greater than placebo for valproate.
Editors note: These data in very young children (aged 3 to 7) resemble other controlled data in the literature about the treatment of older children and adolescents, indicating a superiority of atypical antipsychotics over placebo and a greater magnitude of effect achieved with atypicals than with valproate. Based on these new data and the Federal Drug Administration (FDA) approval of several atypical antipsychotics for children with bipolar illness from ages 10 to 17, Dr. Kowatch recommended a new treatment algorithm for childhood onset bipolar disorder. Read more
RTMS Versus ECT
 We’ve been posting about repeated transcranial magnetic stimulation (rTMS), a treatment in which a magnetic field is applied to a patient’s head, where it is able to affect tissue 2-3 centimeters into the brain.
We’ve been posting about repeated transcranial magnetic stimulation (rTMS), a treatment in which a magnetic field is applied to a patient’s head, where it is able to affect tissue 2-3 centimeters into the brain.
In randomized, controlled, head to head comparisons of electroconvulsive therapy (ECT) and repeated transcranial magnetic stimulation (rTMS), ECT often proves superior, particularly for a more severe psychotic or medically compromised patient. Nonetheless, for a more routine patient with non-psychotic depression, multiple studies have shown approximate equivalence of rTMS and ECT, while rTMS may have a superior benefit-to-risk ratio because it brings less risk of cognitive dysfunction. Thus, given almost equivalent efficacy for some subgroups of depressed patients, and a clear advantage for rTMS in terms of lack of cognitive side effects, this editor/clinician would recommend a course of rTMS prior to a series of ECT for the subgroup of non-emergency treatment-resistant depressed patients.
However, for highly treatment-refractory, severe, and particularly psychotic depression, few treatments appear to approach the efficacy and rapid response onset of electroconvulsive therapy (ECT). ECT as given in the modern era involves anesthesia and muscle paralysis while the patient is mechanically ventilated in order to prevent any damage from muscle contractions during the seizure. Read more
Ziprasidone Improves Mood With Possible Weight Loss Side Effects
 In an open study of bipolar disorder treatment, Shefali Srivastava, Terence Ketter and colleagues at Stanford University evaluated ziprasidone as an aid to patients unresponsive to other medications. This study was part of the multi-center research program Systematic Treatment and Evaluation Program for Bipolar Disorder, or STEP-BD. During naturalistic treatment, ziprasidone was added to an average of 3.6 other psychotropic medications and 1.2 other nonpsychotropic medications patients had already been prescribed. The researchers found substantial improvement in mood with ziprasidone, particularly in the patients who had symptomatic levels of depression at baseline. The research team also observed a mean weight decrease from 195 + 50lbs at baseline to 183 + 47lbs at the final visit, with 34.3% of the patients achieving at least a 7% weight loss with ziprasidone.
In an open study of bipolar disorder treatment, Shefali Srivastava, Terence Ketter and colleagues at Stanford University evaluated ziprasidone as an aid to patients unresponsive to other medications. This study was part of the multi-center research program Systematic Treatment and Evaluation Program for Bipolar Disorder, or STEP-BD. During naturalistic treatment, ziprasidone was added to an average of 3.6 other psychotropic medications and 1.2 other nonpsychotropic medications patients had already been prescribed. The researchers found substantial improvement in mood with ziprasidone, particularly in the patients who had symptomatic levels of depression at baseline. The research team also observed a mean weight decrease from 195 + 50lbs at baseline to 183 + 47lbs at the final visit, with 34.3% of the patients achieving at least a 7% weight loss with ziprasidone.
Mean trial duration was 860 + 700 days, with no subsequent psychotropic agents added in 51.2% of the patients who had a mean trial duration of 221 + 272 days. Ziprasidone was discontinued in 57.3% of the 82 trials after a mean of 208 + 364 days. This was due to side effects in 26.8% of the participants and due to inefficacy for mood in 23.2%.
The investigators concluded that in bipolar patients treated naturalistically with complex pharmacotherapy, ziprasidone decreased overall bipolar illness severity, was helpful in patients with substantial depression at baseline, and also yielded clinically significant weight loss in about one-third of the patients.
Editor’s note: These data are notable because they support ziprasidone’s pattern of weight neutrality and because of the overall improvement in mood symptomatology the drug brought about. Read more
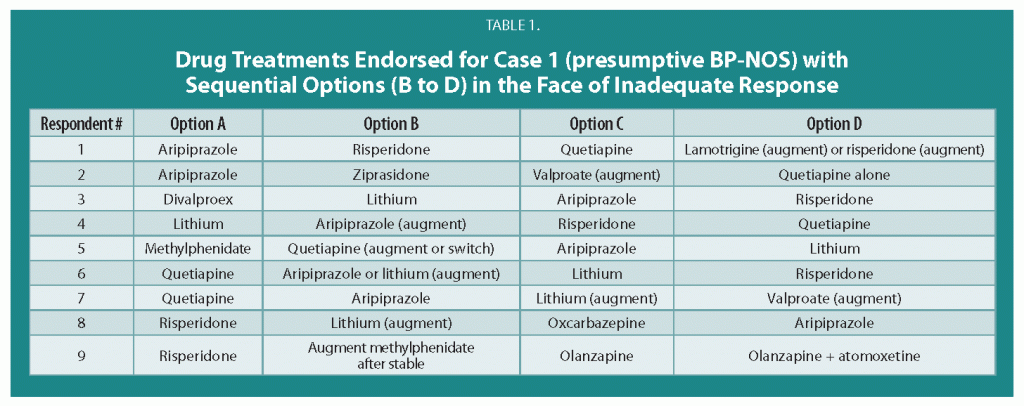
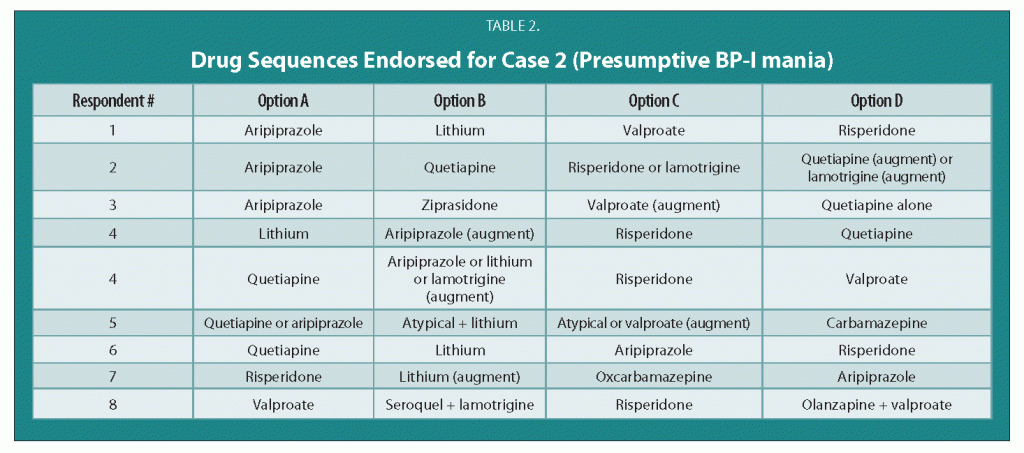
Treatment Guidelines for Two Hypothetical Cases in Children
There are no FDA-approved treatments for children under age 10 with bipolar disorder. For an article in Psychiatric Annals, this editor and Janet Wozniak asked experts how they would sequence treatment of a hypothetical case of a 6-year-old with extreme mood instability consistent with a diagnosis of BP -NOS (see Table I). We also asked how the experts would treat a different case of a 9-year-old with a full-blown psychotic BP-I mania (see Table II).
The results are presented and discussed in detail in the article, and are presented here to reinforce several points. The recommendations for children under 10 and for BP NOS are highly similar to consensus guidelines for older BP I children compiled by Kowatch et al.
Treatments in the face of non-response to option A or others are sequenced differently by different experts, but almost always involve an atypical antipsychotic (AA) or a mood stabilizer (MS) such as lithium, valproate, carbamazepine/oxcarbazepine, or rarely, lamotrigine. Revisions of atypical antipsychotics and mood stabilizers and use of combinations are the common next strategies.
One Expert’s Personal Treatment Algorithm for Bipolar Disorder in Young Children
EDITOR’S NOTE: Dr. Gagin Joshi of Massachusetts General Hospital, who presented the work on carbamazepine and lamotrigine on page 1 provided us with his own general treatment algorithm for youngsters with bipolar disorder.

Omega-3 Fatty Acids (photo from ironmagazine.com)
Joshi typically starts with 0.5 to 2 gms of omega-3 fatty acids because of their benign side-effects profile, the many studies suggesting they are effective in adult mood disorders, and a recent article indicating that they were effective in preventing the conversion of prodromal schizophrenia into full-blown illness in a randomized double-blind controlled study in Australia.
After the omega-3 fatty acids, Joshi’s second choice is typically the atypical antipsychotic aripiprazole (Abilify) because of its lesser degree of weight gain compared to atypicals quetiapine (Seroquel) or risperidone (Risperidol). Risperidone can be a third option if aripiprazole is not effective or tolerated.