Bipolar I patient show dramatic reductions in white matter integrity
Thiel et al in Neuropsychopharmacology (2024) reported that “Compared with HC [healthy controls], BD-I patients exhibited lower FA [fractional anisotropy] in widespread clusters (ptfce-FWE?< 0.001), including almost all major projection, association, and commissural fiber tracts. BD-II patients also demonstrated lower FA compared with HC, although less pronounced (ptfce-FWE?=?0.049).”
Editors Note: These data once more emphasize the importance of using lithium (Li) in bipolar disorder as it can ameliorate the deficits in white matter integrity that are so prominent in the illness. Li also improve the loss of cortical grey matter volume that evolves with illness progression. Li prevents episodes of depression and mania and reduces the incidence of suicides. That Li can reverse or ameliorate brain abnormalities in bipolar disorder is one more piece of evidence that Li should be considered a disease modifying drug (DMD) and started early in the course of illness in almost all bipolar patients. The new mantra for patients and clinicians is: Use more lithium and prevent illness progression.
Changes in brain structure in remitted bipolar patients
Macoveanu et al reported in the Journal of Affective Disorders (2023) that compared to controls that remitted bipolar patients had “a decline in total white matter volume over time and they had a larger amygdala volume, both at baseline and at follow-up time. Patients further showed lower cognitive performance at both times of investigation with no significant change over time….Cognitive impairment and amygdala enlargement may represent stable markers of BD early in the course of illness, whereas subtle white matter decline may result from illness progression.”
Both Obesity and Bipolar Disorders in 2249 Individuals Show White Matter Microstructure Abnormalities
Lorielle Dietze of Dalhousie University “obtained body mass index (BMI) and diffusion tensor imaging derived fractional anisotropy (FA) values from 930 individuals with bipolar disorders (BD), and 1319 control individuals from 20 cohorts in the ENIGMA-BD Working Group.”
They “found that lower FA was associated with both BD and BMI, in five white matter tracts, including the corpus callosum and thalamic radiation. Nine ROIs were correlated with only BD, while higher BMI was uniquely correlated with lower FA in four white matter ROIs.”
They concluded: “For the first time we showed that both obesity and BD demonstrated lower FA in some of the same regions. The impact of obesity may be greater in some tracts in BD individuals.”
Cognitive Function and White Matter Integrity in Individuals With Bipolar Disorder
Highlights from Posters Presented at the Society of Biological Psychiatry Meeting, April 27-29, 2023 in San Diego
Jennifer McDowell reported that they found “significantly reduced FA (fractional anisotropy) values in 85 bipolar probands compared to 66 controls” in multiple (n=8) white matter tracts. There were significantly lower scores in bipolar probands compared to controls on composite scores, ( p = 0.007), verbal fluency, ( p < 0.001), and symbol coding, (p = 0.023). They concluded that: “ Impacted connectivity in critical fiber tracts may be key to understanding the neural underpinnings of deficits, like cognition, observed in this clinical population.”
Editors note: It is of interest that lithium has been shown to normalize some white matter abnormalities in youngsters and help preserve cognitive function in older individuals. On this and many other accounts, way too little lithium is being used in the treatment of patients with bipolar disorder. Lithium not only increases neurogenesis (new grey matter neurons) and hippocampal volume, but also has positive effects on white matter tracts and even increases the length of one’s telomeres (which keeps you more healthy). In other ungrammatical words, “If your brain is not connected right, it don’t work right.”
A NEW VIEW ON MEMORY TO REMEMBER
Steven Ramirez PhD of Boston University gave a talk (8/9/22) on memory for the BBRF hosted by Jeffrey Borenstein President & CEO of the Brain & Behavior Research Foundation. Ramirez showed that positive (food) and negative (shock) memories of different places were stored in different neurons of the hippocampus. If he turned on the positive memories with optical stimulation while a mouse was in a negative memory place and would ordinarily show freezing representing fear behavior, much less freezing occurred with the insertion of the positive memory. Positive memories appear to trump negative memories.
Ramirez found score of genes were activated or turned off in the positive memory cells, some of which, but not all, overlapped with the negative memory cells. Remarkably, the bulk of the unique positive memory genes were related to synaptogenesis and neuroprotection, while the bulk of genes unique to the negative memory cells were related cell death and other toxic factors. Ramirez hopes these data will provide clues to not only helping people with PTSD, but also ultimately providing targets for providing protection against degenerative diseases.
When Ramirez was asked by Borenstein what people could do now, he related his own experience of every morning filling out a form for gratitude and gratefulness for at least 3 things he could be grateful for the previous day or anticipated for the current day. The positive memories that this invoked in him set up his positive and optimistic attitudes for the rest of the day. He recommends this approach of positive memories modulating the current pervasive stressors of the day. For people interested in the details of his experiments summarized above, they should look for the in press articles of Grella et al Nature Communications and Shpokayte et al Nature Communications.
Surface Area of Cortex Is Reduced After Multiple Manic Episodes
In a 2020 article in the journal Psychiatric Research: Neuroimaging, researcher Rashmin Achalia and colleagues described a study of structural magnetic resonance imaging (MRI) that compared 30 people with bipolar I disorder who had had one or several episodes of mania to healthy volunteers. Compared to the healthy volunteers, people with bipolar disorder had “significantly lower surface area in bilateral cuneus, right postcentral gyrus, and rostral middle frontal gyri; and lower cortical volume in the left middle temporal gyrus, right postcentral gyrus, and right cuneus.”
The surface area of the cortex in patients with bipolar I disorder who had had a single episode of mania resembled that of the healthy volunteers, while those who had had multiple manic episodes had less cortical surface area.
The data suggest that compared to healthy volunteers, people with bipolar disorder have major losses in brain surface area after multiple episodes that are not seen in first episode patients. In addition, the researchers found that both the number of episodes and the duration of illness was correlated with the degree of deficit in the thickness in the left superior frontal gyrus. These decreases in brain measures occurred after an average of only 5.6 years of illness.
Editor’s Note: These data once again emphasize the importance of preventing illness recurrence from the outset, meaning after the first episode. Preventing episodes may prevent the loss of brain surface and thickness.
Clinical data has also shown that multiple episodes are associated with personal pain and distress, dysfunction, social and economic losses, cognitive deficits, treatment resistance, and multiple medical and psychiatric comorbidities. These and other data indicate that treatment after a first episode must be more intensive, multimodal, and continuous and include expert psychopharmacological and psychosocial support, as well as family education and support. Intensive treatment like this can be life-saving. The current study also supports the mantra we have espoused: prevent episodes, protect the brain and the person.
Left Prefrontal Strokes Linked to Depression

In a 2021 article in the journal Stroke, researcher Julian Klingbeil and colleagues reported that left, but not right, ventrolateral prefrontal stroke lesions were associated with increased risk of depression at six months post-stroke.
The study included 270 participants who had their first-ever stroke. Six months following their strokes, 19.6% of the participants had depression. Those who scored higher on a scale of depression and anxiety symptoms in the first month after their stroke were more likely to have depression six months after the stroke.
The researchers identified a cluster of locations for stroke lesions, mostly within the left ventrolateral prefrontal cortex, that they linked to depression symptoms six months post-stroke. Klingbeil and colleagues hope that recognizing lesions in this region as risk factors for depression will help with early diagnosis of depression among people who recently had a stroke.
Editor’s Note: Antidepressants have been shown to improve post-stroke recovery of neurological functional (and depression) that is caused by the cutoff of blood supply during a stroke (ischemia). Patients and their family members should talk with their neurologist about treatment of ischemic strokes with antidepressants, especially when the lesions occur on the left side of the brain.
White Matter Disturbances in Bipolar Disorder
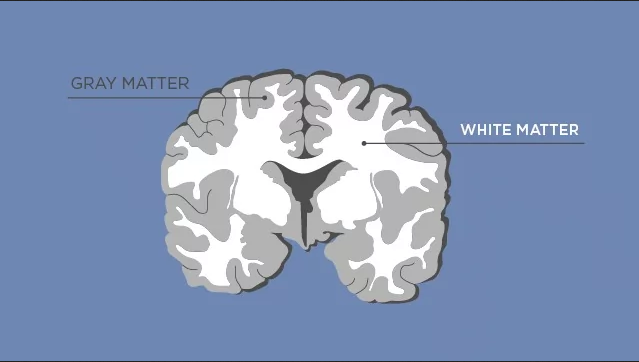
At the 2020 meeting of the International Society for Bipolar Disorders, researcher Clare Beasley described the cellular and molecular underpinnings of the white matter abnormalities typically seen in children and adults with bipolar disorder. Researchers consistently see white matter abnormalities in neuroimaging studies of bipolar disorder, but not much is understood about what creates these deficits.
Beasley and colleagues studied autopsy specimens and found that compared to controls, people with bipolar disorder had a number of abnormalities affecting glial cells, lipid composition, and axons.
The researchers found increased density of oligodendrocytes (glial cells that produce the myelin that wraps around axons, the long fibers of nerve cells where impulses travel out to other cells) and an associated protein called CNP in the prefrontal cortex. The myelin is what makes up white matter, while gray matter consists of cell bodies of neurons and glial cells.
People with bipolar disorder also had differently-shaped astrocytes, another type of glial cell that abuts synapses. The researchers found changes in lipid composition, including phospholipid and fatty acid levels, in the white matter of people with bipolar disorder. There were also problems with axons. Beasley and colleagues noted lower density of axon-associated proteins, which are involved in transport of substances along the axons in people with bipolar disorder.
The authors conclude that these data implicate specific disturbances in oligodendrocytes and axonal function associated with the white matter alterations usually seen in neuroimages of people with bipolar disorder.
Translocator Protein Levels in Brain Predict Response to Anti-Inflammatory Celecoxib in Major Depressive Disorder
Gliosis describes changes in glia that result from damage to the central nervous system. Researchers can use PET scans (positron emission tomography) to measure the extent of gliosis in the brain. But a new study explored whether these PET scans could instead be used to determine who might respond to a given medication.
Researcher Sophia Attwells and colleagues reported in the journal Biological Psychiatry in 2020 that people with high levels of translocator protein (TSPO), a measure of gliosis and inflammation, had a better antidepressant response to the anti-inflammatory drug celecoxib than patients who started out with lower levels of TSPO.
The study participants, who had treatment-resistant depression, all received 200mg of the anti-inflammatory drug celecoxib twice/day for eight weeks on an open (non-blind) basis. Before they began taking celecoxib, the participants received PET scans to measure translocator protein total distribution volume (TSPO VT) in the prefrontal cortex and the anterior cingulate cortex.
Patients with high levels of TSPO showed greater reductions in depression ratings over the course of the study than those with normal levels of TSPO at baseline.
Attwells and colleagues conclude that “this personalized medicine approach of matching a marker of gliosis to [an anti-inflammatory treatment] …should be applied in early development of novel therapeutics, in particular for [treatment-resistant depression].”
Editor’s Note: These findings are of considerable importance, as they are among the first to indicate that measures of inflammation may predict response to an anti-inflammatory medication such as celecoxib. In a 2013 article in the journal JAMA Psychiatry, Charles L. Raison and colleagues reported that patients with high levels of the peripheral inflammatory marker CRP saw marked improvement in their depression when they received the anti-inflammatory treatment infliximab while those with lower or normal levels of inflammation actually worsened.
Quetiapine Reduced Childhood Mania, Especially in Those with Thicker Frontal Temporal Regions
 In a symposium at the 2019 meeting of the American Academy of Child and Adolescent Psychiatry, researcher Melissa P. Delbello reported that six weeks of treatment with either lithium or quetiapine was effective in childhood mania, but quetiapine had a higher response rate of 71% versus 46% for lithium. Delbello found two types of structural changes on functional magnetic resonance imaging (fMRI). Some children had thicker frontal temporal regions, while others had thinning in these areas. The first group of patients had a 100% response to quetiapine, but only 53% of the second group responded to quetiapine.
In a symposium at the 2019 meeting of the American Academy of Child and Adolescent Psychiatry, researcher Melissa P. Delbello reported that six weeks of treatment with either lithium or quetiapine was effective in childhood mania, but quetiapine had a higher response rate of 71% versus 46% for lithium. Delbello found two types of structural changes on functional magnetic resonance imaging (fMRI). Some children had thicker frontal temporal regions, while others had thinning in these areas. The first group of patients had a 100% response to quetiapine, but only 53% of the second group responded to quetiapine.
In contrast, other researchers have found lithium superior to quetiapine. Vivian Kafantaris showed that patients who respond well to lithium show improvements in white matter abnormalities. Michael Berk and colleagues found that a year on lithium was superior to quetiapine on all measures including cognition and brain imaging in patients having their first episode of mania.

