Risk of Attempted or Completed Suicide in Borderline Personality Disorder: Reduced with ADHD meds; Increased with Benzodiazepines
Johannes Lieslehto et al 2023 reported in JAMA New. Open on the comparative effectiveness in 22,601 individuals with BPD that “ADHD medication was the only pharmacological treatment associated with reduced risk of suicidal behavior among patients with BPD. Conversely, the findings suggest that benzodiazepines should be used with care among patients with BPD due to their association with increased risk of suicide.” Mood stabilizers had no effect while antipsychotics minimally and antidepressants moderately increased risk of suicide attempts or completed suicide.
U.S. FDA Approves VRAYLAR® (cariprazine) as an Adjunctive Treatment for Major Depressive Disorder
“A Phase 3 Study 3111-301-001 showed a clinically and statistically significant change from baseline to week six in the Montgomery-Åsberg Depression Rating Scale (MADRS) total score for patients treated with cariprazine at 1.5 mg/day + ADT compared with placebo + ADT. A second registration-enabling study, RGH-MD-75, showed a clinically and statistically significant change from baseline to week eight in the MADRS total score for patients treated with cariprazine at 2-4.5 mg/day (mean dose 2.6 mg) + ADT compared with placebo + ADT.
Cariprazine was generally well tolerated in 6- and 8-week studies. Mean weight change was < 2lbs and ? 3% of patients had a weight increase of ? 7%.
The starting dosage of VRAYLAR is 1.5 mg once daily. Depending upon clinical response and tolerability, the dosage can be increased to 3 mg once daily on Day 15. In clinical trials, dosage titration at intervals of less than 14 days resulted in a higher incidence of adverse reactions. The maximum recommended dosage is 3 mg once daily.
Most common adverse reactions observed in the adjunctive MDD studies (? 5% and at least twice the rate of placebo) were:
Akathisia, nausea, and insomnia at the recommended doses in 6-week, fixed-dose trials
Akathisia, restlessness, fatigue, constipation, nausea, increased appetite, dizziness, insomnia, and extrapyramidal symptoms in one 8-week flexible-dose trial at a titration of less than 14 days”
In Phase 3 Clinical Trials, Antipsychotic Treatment Lumateperone Is Found Effective in Bipolar Depression
Lumateperone is an antipsychotic medication that is currently approved by the US Food and Drug Administration for the treatment of schizophrenia (under the trade name Caplyta). New studies suggest that the drug is also effective in bipolar I and bipolar II depression.
Lumateperone modulates the activity of the neurotransmitters dopamine, serotonin, and glutamate. It modulates D1 and D2 dopamine receptors, partially activating presynaptic receptors while partially blocking postsynaptic receptors. Lumateperone acts as an antagonist blocking serotonin 5-HT2A receptors, and it augments activity at both NMDA and AMPA glutamate receptors.
Because of lumateperone’s complex pharmacology, it is not clear which of these activities are primarily responsible for its antidepressant and antipsychotic activities.
New research presented at the 2020 meeting of the International Society for Bipolar Disorders showed that lumateperone reduces bipolar depression.
Researcher Susan Kozauer presented research from a six-week study of 377 patients who were randomized to receive treatment with either 42mg of lumateperone (n=188), taken once daily in the evening, or placebo (n=189). The patients had been diagnosed with bipolar I or II disorder and were experiencing an episode of major depression.
Patients taking lumateperone saw significantly greater improvement in depression than those in the placebo group. Among those taking lumateperone, 60% of those who were markedly ill or worse at baseline improved to mildly ill or better, compared to 43% of those taking placebo.
Researcher Suresh Durgam described improvements in specific symptoms that make up the Montgomery-Åsberg Depression Rating Scale (MADRS) in those patients who received lumateperone. The greatest improvements compared to placebo were in sadness, inner tension, and reduced sleep. By the 29th day of the study, 8 of 10 items on the scale had improved significantly compared to placebo, and all items had improved by day 43.
The side-effects profile of lumateperone was presented by researcher Lakshmi Yatham.
Among those taking lumateperone, 8.5% experienced sleepiness compared to 1.1% of those in the placebo group, while 6.4% of the lumateperone group experienced nausea compared to 2.1% of the placebo group. Most effects were mild or moderate in severity. Changes in weight, metabolic measures, extrapyramidal motor effects, and prolactin were minimal in both the lumateperone group and the placebo group.
Editor’s Note: Lumateperone (Caplyta) joins a list of other atypicals that are efficacious in bipolar depression. These include olanzapine-fluoxetine (Symbyax), quetiapine (Seroquel), lurasidone (Latuda), and cariprazine (Vraylar). Lumateperone is currently only FDA-approved for schizophrenia, but approval for bipolar depression should be rapidly forthcoming based on the data presented at the ISBD meeting.
Bipolar depression used to be an orphan diagnosis, with few efficacious treatments. This is now beginning to change, and treating patients with bipolar disorder using antidepressants designed to treat unipolar depression (for which there is little evidence of efficacy) should begin to recede.
New Type of Antipsychotic Drug for Schizophrenia Looks Promising
In a 2020 article in the New England Journal of Medicine, researcher Kenneth S. Koblan and colleagues described a new type of antipsychotic drug treatment for schizophrenia. Almost all other antipsychotic drugs block dopamine D2 receptors, while atypical antipsychotics also block the serotonin 5HT2 receptor. They are described as antagonists at these receptors.
In contrast, the new drug is an agonist or activator of two different receptors. The drug SEP-363856 (also called SEP-856) activates the trace amine–associated receptor 1 (TAAR1) and 5-hydroxytryptamine (or serotonin) type 1A (5-HT1A) receptors.
Blocking D2 receptors can cause Parkinson’s-like symptoms (such as tremor, masked faces, and impaired movement or speech) and other extrapyramidal side effects (such as slurred speech, slow movements, or restless legs.) In contrast, SEP-856 seems to have a better side effects profile than these types of drugs while also being highly effective.
Patients with an acute exacerbation of schizophrenia were assigned to receive either placebo or once-daily treatment with SEP-856 (either 50mg or 75mg) for four weeks. A total of 120 patients received SEP-856 while 125 received placebo.
Compared to the placebo group, the SEP-856 group showed significantly greater reductions on a scale of positive and negative symptoms of schizophrenia by the end of the four weeks. Side effects included some sleepiness and gastrointestinal symptoms, but the incidence of extrapyramidal symptoms and changes in the levels of lipids, glycated hemoglobin, and prolactin were similar in both groups. There was one sudden death from cardiac causes in the SEP-856 group, which was not thought to be drug-related.
Editor’s Note: This drug acting on trace amine–associated receptor 1 (TAAR1) and 5HT1A receptors could herald a new and better tolerated type of antipsychotic. It is also being studied for psychosis in Parkinson’s disease. Since all of the antipsychotics that treat schizophrenia have also shown antimanic efficacy, we look forward to future studies of this unique drug in patients with mania.
Cannabidiol May Help Treat Schizophrenia
 A 2017 article by researcher Philip McGuire and colleagues in the American Journal of Psychiatry reports that when added to antipsychotic medication, cannabidiol, a component of marijuana, improved positive symptoms of schizophrenia, such as hallucinations and delusions, more than did the addition of a placebo.
A 2017 article by researcher Philip McGuire and colleagues in the American Journal of Psychiatry reports that when added to antipsychotic medication, cannabidiol, a component of marijuana, improved positive symptoms of schizophrenia, such as hallucinations and delusions, more than did the addition of a placebo.
In the double-blind, parallel-group study, 43 participants received 1000 mg/day of cannabidiol in addition to their regular antipsychotic medication, while 45 participants received a placebo alongside their regular medication.
Side effects were minimal, and after six weeks those who received cannabidiol had decreased positive symptoms and were more likely to be considered improved and not severely unwell.
Editor’s Note: It is important to emphasize that cannabidiol is only a minor component of marijuana, which contains much more tetrahydrocannabinol (THC), which is psycho-mimetic, i.e. it can worsen psychosis. Pure cannabidiol is not readily available to the public.
20-Year Study Finds Clozapine and Long-Acting Injectable Antipsychotics Most Effective at Preventing Re-Hospitalizations for Schizophrenia
Few studies have evaluated the comparative long-term effectiveness of antipsychotics in preventing relapse, but a 2017 study from Finland published in the journal Schizophrenia Bulletin by Heidi Taipale and colleagues did just that, and found that clozapine and long-acting injectable antipsychotic drugs were most effective at preventing psychiatric re-hospitalizations.
The Finnish health care registry was used to prospectively collect data on the treatment of every person who received inpatient care for schizophrenia between 1972 and 2014. The patients totaled 62,250 including 8,719 in their first episode of schizophrenia. Follow-up to evaluate antipsychotic use began at 1996 for those with ongoing treatment, and upon first discharge from the hospital for those patients in their first episode. The follow-up time ranged from 6.9 to 20 years with an average of 14.1 years. During the follow-up period, 59% of patients were readmitted to psychiatric inpatient care.
Among the drugs with the lowest rates of relapse, olanzapine long-acting injection, clozapine, and paliperidone long-acting injection were associated with the least risk of psychiatric re-hospitalization. Among patients in a first episode, taking flupentixol long-acting injection, olanzapine long-acting injection, or perphenazine long-acting injection had the lowest risk of psychiatric re-hospitalization. Clozapine and the long-acting injections also had the least risk of hospitalization for any cause.
Large Finnish Study Finds Lithium is Best at Preventing Re-Hospitalizations in Bipolar Disorder
A 2018 article in the journal JAMA Psychiatry reports that lithium and long-acting antipsychotic injections were most effective at preventing re-hospitalizations among people with bipolar disorder.
The study by Markku Lähteenvuo and colleagues included 18,018 Finnish patients with bipolar disorder. A national database contained information on any hospitalizations that occurred among the patients and what medications were dispersed to patients.
Among the participants, 54% (9,721 patients) were re-hospitalized at least once over a study period of 16 years. Medications associated with the smallest risk of re-hospitalization for psychiatric reasons were long-acting injections of risperidone, gabapentin, long-acting injections of perphenazine, and lithium carbonate.
When the researchers looked at hospitalizations for any cause (not just psychiatric illness), lithium was associated with the least risk of re-hospitalization, while benzodiazepines had the greatest risk, both for psychiatric re-hospitalization and re-hospitalization for any cause.
Long-acting injectable medications were associated with less risk of re-hospitalization compared to the identical medications delivered orally.
Lähteenvuo and colleagues concluded, “Lithium…should remain as the first line of treatment for bipolar disorder, after decades of underprescription.” They suggest that long-acting injectable medications may be a good alternative to prevent relapse in patients for whom lithium is unsuitable.
Editor’s Note: In addition to lithium’s ability to prevent depressions and manias, it also increases the volume of the hippocampus and protects against a diagnosis of dementia in old age. Lithium decreases the risk for suicide and also increases the length of telomeres, bits on the ends of DNA strands that protect them as they replicate, which are important to the maintenance of both physical and psychiatric health. When lithium is used cautiously to maintain doses below a given patient’s side effects threshold, it is very well tolerated by most individuals.
Topiramate Plus Antipsychotic Medication Better Than Antipsychotics Alone for Schizophrenia Spectrum Disorders
 A 2016 meta-analysis has shown that the combination of the anticonvulsant topiramate and antipsychotic medication reduces symptoms of schizophrenia spectrum disorders more than antipsychotic medication alone. Researchers led by Christoph U. Correll analyzed the results of eight studies in which the topiramate-antipsychotic combination was compared to antipsychotics alone or with placebo.
A 2016 meta-analysis has shown that the combination of the anticonvulsant topiramate and antipsychotic medication reduces symptoms of schizophrenia spectrum disorders more than antipsychotic medication alone. Researchers led by Christoph U. Correll analyzed the results of eight studies in which the topiramate-antipsychotic combination was compared to antipsychotics alone or with placebo.
The combination of topiramate and antipsychotic medication was superior at reducing general psychopathology, including both negative and positive symptoms of schizophrenia. The combination was also associated with lower body weight and body mass index (BMI) compared to antipsychotics alone.
The studies included in the meta-analysis used a variety of antipsychotic medications. When these were compared, the combination of topiramate and clozapine was more effective than other combinations at reducing psychopathology. However, the combination of topiramate and clozapine was also associated with less weight loss than combinations using other antipsychotics.
In terms of side effects, topiramate was associated with more paresthesia (a burning or prickling sensation, often in the hands or feet) than placebo.
The study was published in the Journal of Clinical Psychiatry.
Antipsychotic Use During Pregnancy Most Likely Safe
A new study suggests that women can continue using antipsychotic medications during the first trimester of pregnancy without meaningfully increasing the risk of birth defects in their offspring.
The study, by Krista F. Huybrechts and colleagues in the journal JAMA Psychiatry, looked at Medicaid users who filled at least one prescription for an antipsychotic medication during their first trimester of pregnancy, when an embryo’s vital organs are formed, and went on to have a live birth. Birth defects, including cardiac malformations, in these children were identified in the first 90 days after delivery and compared to the number of such abnormalities in the children of women on Medicaid who did not receive a prescription for an antipsychotic drug during the first trimester of pregnancy. The number of abnormalities was slightly higher in the children of women who had received atypical antipsychotics than in those who had not, and slightly lower in the children of women who had received a typical antipsychotic than in those who had not.
Huybrechts and colleagues concluded that taking an antipsychotic medication during the first trimester of pregnancy does not meaningfully increase the risk of birth defects in the offspring.
The children of women who took the antipsychotic risperidone did have a small increased risk of birth defects, including cardiac malformations. The researchers called for additional study of risperidone use during pregnancy.
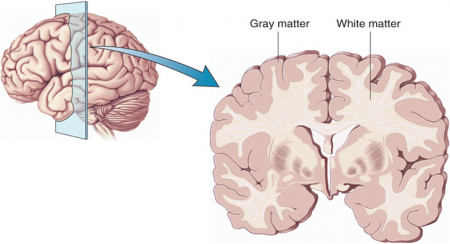
Atypical Antipsychotics May Slow Loss of Gray Matter in Schizophrenia
Progressive losses in gray matter have been observed in the cortex of people with schizophrenia, and those at high risk for the illness. In the past, studies have shown that the amount of antipsychotics a patient is exposed to is correlated with the extent of their deficits in gray matter, suggesting that antipsychotic treatment could exacerbate gray matter loss.
A new meta-analysis by Antotonio Vita and colleagues in the journal Biological Psychiatry shows that first-generation antipsychotics were associated with greater losses in gray matter compared with atypical antipsychotics, which seemed to slow the loss of gray matter.
The meta-analysis analyzed data from 18 longitudinal studies comparing a total of 1155 patients with schizophrenia to 911 healthy control participants. Magnetic resonance imaging (MRI) scans showed that over time, patients with schizophrenia lost more cortical gray matter volume. The patients’ cumulative intake of any kind of antipsychotic between MRI scans was associated with gray matter losses. But when Vita and colleagues drilled down to find differences between patients taking first-generation antipsychotics and those taking second-generation atypical antipsychotics, they found that patients with higher average daily intake of first-generation antipsychotics had greater losses in gray matter, while patients with higher average daily intake of atypical antipsychotics had less progressive losses in gray matter.
This study is the first to compare the effects of first-generation antipsychotics, which were developed in the 1960s, with those of atypical antipsychotics, which came into frequent use in the late 1980s, on cortical gray matter loss in schizophrenia. While first-generation antipsychotics are associated with the side effect of tardive dyskinesia, involuntary movements of the face and jaw, atypical antipsychotics are most commonly associated with weight gain.
Three studies have randomly assigned patients with schizophrenia to receive either first-generation or atypical antipsychotics. In these studies as well, second-generation antipsychotics were associated with smaller losses in gray matter.
The authors speculate that either second-generation antipsychotics may have neuroprotective effects, or first-generation antipsychotics may have neurotoxic effects. They also suggest that first-generation antipsychotics may not have the capacity to interfere with the natural progression of schizophrenia in terms of gray matter losses.
Future studies may investigate differences between specific antipsychotic medications’ effects on gray matter volume. Vita and colleagues reported that in the analysis, the atypical antipsychotic clozapine was associated with the least loss of gray matter of any medication in the included studies.
Editor’s Note: This study is important because it adds to findings questioning the conclusions of a large National Institute of Mental Health–sponsored study known as CATIE and a meta-analysis by John Geddes published in the journal BMJ in 2000, in which he wrote that “There is no clear evidence that atypical antipsychotics are more effective or better tolerated than conventional (first generation) antipsychotics.” Read more