Environment Can Leave “Molecular Scars” Via Epigenetic Processes

A 2020 review article by researchers Julia Richetto and Urs Meyer in the journal Biological Psychiatry provides a good overview of the role epigenetic modifications play in schizophrenia and related disorders.
The article provides a powerful understanding of how the environment can induce long-lasting changes in the structure of DNA (not only in schizophrenia, but also in bipolar disorder). This process, known as epigenetics, can have life-long influences on brain chemistry and behavior, and remarkably, some of these epigenetic changes can even be transmitted to the next generation.
While the sequence of DNA that one inherits from one’s parents does not change over the course of one’s life, what can change is how loosely or tightly the DNA is wound around proteins called histones, making it easier or harder to transcribe the genetic material held there. The addition of a methyl group to DNA usually inhibits transcription, while the addition of an acetyl group to histones usually facilitates transcription. These alterations in the shape of the DNA that result from environmental exposures or behavior can be passed on through generations.
Richetto and Meyer describe these chemical changes to DNA as “molecular scars,” which are left when environmental stress occurs during sensitive developmental periods. For example, patients with schizophrenia who experienced stressors in early life have higher levels of the enzyme histone deacetylase than patients who had stress or trauma later in life. Histone deacetylase would remove the acetyl groups on histones, which would inhibit gene transcription.
Other factors that have been implicated in epigenetic modifications in schizophrenia, such as DNA methylation of key developmental pathways, include pre- or post-natal stress, a challenge to a mother’s immune system during pregnancy, pre- and post-natal nutrition, exposure to drugs or toxic substances, and cannabis use in adolescence.
Richetto and Meyer suggest that epigenetics may explain why schizophrenia (and we would add bipolar disorder) can differ so much across individuals, and may help researchers and clinicians determine how best to treat different individuals.
Editor’s Note: This editor has written about how epigenetic changes can mediate sensitization to the recurrence of life stressors, episodes of mood disorder, and bouts of substance abuse, each of which can drive illness exacerbation and progression in bipolar disorder (see the 2016 article by Robert M. Post in the journal Bipolar Disorders, “Epigenetic basis of sensitization to stress, affective episodes, and stimulants: implications for illness progression and prevention”).
The chemical changes to our DNA, histones, and microRNA emphasize how important it is to begin long-term preventative treatment starting after a first episode of mania. This not only helps limit episode recurrence and the accumulation of stressors and bouts of substance use that can cause illness deterioration, but also limit the placement of these “molecular scars” on our DNA. The key to treating bipolar disorder is: prevent episodes, protect the person and the brain.
New Type of Antipsychotic Drug for Schizophrenia Looks Promising
In a 2020 article in the New England Journal of Medicine, researcher Kenneth S. Koblan and colleagues described a new type of antipsychotic drug treatment for schizophrenia. Almost all other antipsychotic drugs block dopamine D2 receptors, while atypical antipsychotics also block the serotonin 5HT2 receptor. They are described as antagonists at these receptors.
In contrast, the new drug is an agonist or activator of two different receptors. The drug SEP-363856 (also called SEP-856) activates the trace amine–associated receptor 1 (TAAR1) and 5-hydroxytryptamine (or serotonin) type 1A (5-HT1A) receptors.
Blocking D2 receptors can cause Parkinson’s-like symptoms (such as tremor, masked faces, and impaired movement or speech) and other extrapyramidal side effects (such as slurred speech, slow movements, or restless legs.) In contrast, SEP-856 seems to have a better side effects profile than these types of drugs while also being highly effective.
Patients with an acute exacerbation of schizophrenia were assigned to receive either placebo or once-daily treatment with SEP-856 (either 50mg or 75mg) for four weeks. A total of 120 patients received SEP-856 while 125 received placebo.
Compared to the placebo group, the SEP-856 group showed significantly greater reductions on a scale of positive and negative symptoms of schizophrenia by the end of the four weeks. Side effects included some sleepiness and gastrointestinal symptoms, but the incidence of extrapyramidal symptoms and changes in the levels of lipids, glycated hemoglobin, and prolactin were similar in both groups. There was one sudden death from cardiac causes in the SEP-856 group, which was not thought to be drug-related.
Editor’s Note: This drug acting on trace amine–associated receptor 1 (TAAR1) and 5HT1A receptors could herald a new and better tolerated type of antipsychotic. It is also being studied for psychosis in Parkinson’s disease. Since all of the antipsychotics that treat schizophrenia have also shown antimanic efficacy, we look forward to future studies of this unique drug in patients with mania.
Endocannabinoid System May Help Explain Tourette Syndrome

Endocannabinoids are neurotransmitters produced by the human body that attach to cannabinoid receptors in the brain, the same receptors that are affected by the consumption of cannabis products.
Tourette syndrome, a neurodevelopmental disorder characterized by tics and psychological symptoms, is probably caused by some dysfunction involving the neurotransmitter dopamine. The syndrome is usually treated with dopamine receptor blockers but is also eased by cannabis use and treatment with THC, the main psychoactive component in cannabis. Recently, researchers set out to determine whether concentrations of endocannabinoids in the cerebrospinal system are related to Tourette syndrome.
In an article published in the journal Neuropsychopharmacology in 2020, researcher Kirsten R. Müller-Vahl and colleagues report that endocannabinoid concentrations were significantly higher in the cerebrospinal fluid of 20 people with Tourette’s syndrome than in 19 control participants without Tourette’s.
The researchers found elevations in the endocannabinoids AEA and 2-AG, the endocannabinoid-like ligand PEA, and the metabolite AA in the participants with Tourette’s syndrome. Levels of 2-AG in the cerebrospinal fluid correlated with severity of attention-deficit hyperactivity disorder symptoms, an aspect of the syndrome.
It is possible that higher concentrations of endocannabinoids are present in the syndrome because they compensate for the overactive influence of dopamine. This explanation would fit with the effectiveness of cannabis in treating Tourette’s. However, that has not yet been determined, and it is also possible that the endocannabinoids are a reaction to dysfunction involving other neurotransmitters, are incidental to the syndrome, or in the best case that they are a direct cause of the syndrome.
Müller-Vahl and colleagues suggest that based on the effectiveness of cannabis in treating Tourette’s, it may turn out that the syndrome is a sort of endocannabinoid deficiency. They believe this hypothesis is not counteracted by the high levels of cannabinoids they found in Tourette’s patients in this study, because these high levels may be accompanied by a reduced number or reduced sensitivity of the cannabinoid receptors or overactivity in the enzymes that break down endocannabinoids, such that it is difficult to maintain normal levels of these neurotransmitters.
Cannabis Withdrawal Syndrome Occurs in Almost Half of Regular Users

A recent systematic review and meta-analysis by researcher Anees Bahji and colleagues in the open access medical journal JAMA Network Open describes the symptoms and prevalence of cannabis withdrawal syndrome.
The most recent edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) indicates that cannabis withdrawal syndrome “requires the presence of at least 3 of the following symptoms developing within 7 days of reduced cannabis use: (1) irritability, anger, or aggression; (2) nervousness or anxiety; (3) sleep disturbance; (4) appetite or weight disturbance; (5) restlessness; (6) depressed mood; and (7) somatic symptoms, such as headaches, sweating, nausea, vomiting, or abdominal pain.”
According to Bahji and colleagues, cannabis withdrawal syndrome occurred in 47% of regular users. Higher rates of withdrawal were found among those in clinical settings, those who also used tobacco or other substances, and those who used cannabis daily.
Bahji and colleagues write that while many people believe that cannabis is relatively harmless, it actually has a variety of associated risks. Short-term risks include impaired short-term memory and motor coordination, altered judgment, paranoia, and psychosis. Long-term effects include addiction, altered brain development, poor educational outcomes, cognitive impairment, diminished quality of life, increased risk of chronic respiratory tract and psychotic disorders, injuries, motor vehicle collisions, and suicide.
The researchers warned that users of cannabis may resume cannabis use to allay the depression and anxiety symptoms that are part of the withdrawal syndrome, perpetuating the long-term withdrawal cycle.
Bahji and colleagues suggest that because of the high prevalence of the withdrawal syndrome, doctors should screen patients for cannabis withdrawal, particularly men and frequent cannabis users. They write, “Clinicians should be aware of CWS as it is associated with clinically significant symptoms, which can trigger resumption of cannabis use and serve as negative reinforcement for relapse during a quit attempt.” Doctors can offer support for those reducing their cannabis consumption.
Fish Oil Monotherapy on Depression in Adolescents at High Risk for Bipolar I Disorder: Ambiguity Persists

Researcher Robert K. McNamara and colleagues reported in the Journal of Child and Adolescent Psychopharmacology in a 2020 article that 12 weeks of treatment with omega-3 fatty acids in the form of fish oil did not reduce depression symptoms in adolescents at risk for bipolar disorder when compared to placebo. The primary outcome measured was the results of the Childhood Depression Rating Scale-Revised (CDRS-R).
Fish oil did perform better than placebo on two parts of the rating scale: symptom severity and symptom improvement, especially in weeks 11 and 12 of the study. Omega-3 fatty acids increased creatine and choline in the anterior cingulate, and also increased polyunsaturated fatty acids in red blood cells. The treatment was safe and well-tolerated.
A total of 42 patients between the ages of 9 and 21 who had been diagnosed with depression and had at least one parent with bipolar I disorder received either placebo or 3 fish oil capsules per day. Each capsule contained 450?mg EPA, 40?mg docosapentaenoic acid (DPA), and 260?mg DHA for a total daily dose of 2130?mg EPA + DHA.
Editor’s Note: Ambiguity persists about whether omega-3 fatty acids can improve unipolar or bipolar depression, attention-deficit hyperactivity disorder (ADHD), or prevent the progression of schizophrenia symptoms to the full syndrome. Given the lack of side effects, and the documented effects on red blood cells and brain choline, clinical use of these compounds could be considered in some circumstances.
Translocator Protein Levels in Brain Predict Response to Anti-Inflammatory Celecoxib in Major Depressive Disorder
Gliosis describes changes in glia that result from damage to the central nervous system. Researchers can use PET scans (positron emission tomography) to measure the extent of gliosis in the brain. But a new study explored whether these PET scans could instead be used to determine who might respond to a given medication.
Researcher Sophia Attwells and colleagues reported in the journal Biological Psychiatry in 2020 that people with high levels of translocator protein (TSPO), a measure of gliosis and inflammation, had a better antidepressant response to the anti-inflammatory drug celecoxib than patients who started out with lower levels of TSPO.
The study participants, who had treatment-resistant depression, all received 200mg of the anti-inflammatory drug celecoxib twice/day for eight weeks on an open (non-blind) basis. Before they began taking celecoxib, the participants received PET scans to measure translocator protein total distribution volume (TSPO VT) in the prefrontal cortex and the anterior cingulate cortex.
Patients with high levels of TSPO showed greater reductions in depression ratings over the course of the study than those with normal levels of TSPO at baseline.
Attwells and colleagues conclude that “this personalized medicine approach of matching a marker of gliosis to [an anti-inflammatory treatment] …should be applied in early development of novel therapeutics, in particular for [treatment-resistant depression].”
Editor’s Note: These findings are of considerable importance, as they are among the first to indicate that measures of inflammation may predict response to an anti-inflammatory medication such as celecoxib. In a 2013 article in the journal JAMA Psychiatry, Charles L. Raison and colleagues reported that patients with high levels of the peripheral inflammatory marker CRP saw marked improvement in their depression when they received the anti-inflammatory treatment infliximab while those with lower or normal levels of inflammation actually worsened.
Gabapentin is Effective in Alcohol Use Disorder in Patients with Alcohol Withdrawal Symptoms

Researcher Raymond F. Anton and colleagues reported in the journal JAMA Internal Medicine that compared with placebo, the anticonvulsant medication gabapentin helped people with alcohol use disorders reduce their drinking or abstain from drinking, especially those who had more withdrawal symptoms before treatment.
Ninety-six participants were randomized to receive either placebo or 1200mg/day of gabapentin for 16 weeks.
In the study, 27% of participants who took gabapentin had no heavy drinking days (compared to 9% among those who took placebo) and 18% achieved total abstinence (compared to 4% among those who took placebo). Gabapentin was most effective in those with a history of alcohol withdrawal symptoms. An impressive 41% of participants with high alcohol withdrawal symptoms who took gabapentin achieved total abstinence compared with 1% of participants in the placebo group.
Gabapentin, which is used to treat epilepsy, influences GABA and glutamate transmitters and inhibits the alpha 2gamma-1 voltage sensitive calcium channel, which is upregulated in chronic alcohol exposure.
Better One-Year Clinical Outcomes After Four Weeks of Theta Burst Stimulation for PTSD Than After Two Weeks
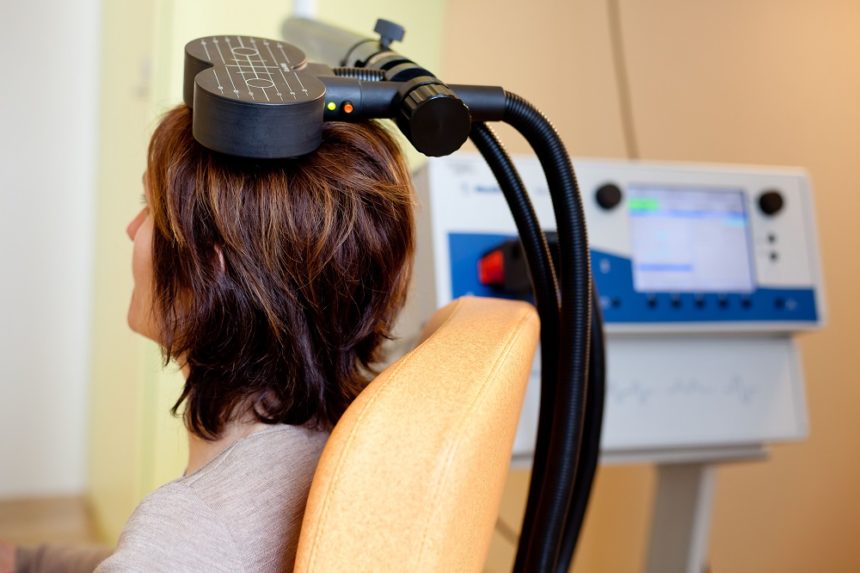
In a 2019 article in the journal Neuropsychopharmacology, Nicholas J. Petrosino and colleagues described findings from one year of follow-up with veterans suffering from post-traumatic stress disorder (PTSD) who received intermittent theta burst transcranial magnetic stimulation (iTBS) in a four-week crossover study.
In the first two weeks of the study, half of the 50 participants (who were mostly male and had an average age of 51) received iTBS while the others were given a sham procedure. Then all the participants received iTBS on an open (non-blind) basis for two more weeks.
At one month, those who had received four total weeks of iTBS had better outcomes than those who had received only two weeks of active iTBS. These results were published in the American Journal of Psychiatry in 2019 in an article by Noah S. Philip and colleagues.
The researchers went on to look at longer-term outcomes, namely time until relapse (a major event such as a re-hospitalization or suicide). After one year, those who received four weeks of iTBS went 9 to 11 months without relapsing (296.0 days ± 22.1), while those who received only two weeks of iTBS went 5 to 7 months before relapsing (182.0 days ± 31.9).
It seems that more iTBS may be better than less iTBS for PTSD in both the short and long term.
Cannabis Use in Adolescence Linked to Depression and Suicidality in Young Adulthood

In a meta-analysis published in the journal JAMA Psychiatry in 2019, researcher Gabriella Gobbi and colleagues analyzed findings from 11 studies including a total of 23,317 participants and found that cannabis use in adolescence (before age 18) was associated with a significantly increased risk of depression, suicidality, and suicide attempts in young adulthood (between 18 and 32 years of age).
The researchers did not find a link between cannabis use and anxiety.
Editor’s Note: Cannabis use is not as harmless as many teens may believe.
In Animal Model, Long-Term THC Exposure Interferes with Cortical Control of the Nucleus Accumbens

In an article in the journal Biological Psychiatry, researchers Eun-Kyung Hwang and Carl R. Lupica reported that in rats, long-term use of THC (tetrahydrocannabinol) weakens input from the cortex to the reward area of the brain, the nucleus accumbens (NAc). Long-term THC use also strengthens connections to the NAc from emotional control (limbic) regions, such as the basolateral amygdala and ventral hippocampus. Hwang and Lupica reason that this shift from cortical control of the NAc to limbic control likely contributes to the cognitive and psychiatric symptoms associated with cannabis use.
Editor’s Note: Street marijuana largely contains THC rather than CBD, the beneficial, anxiety-reducing component of cannabis. Cannabis products are being decriminalized, but it is important to remember that those with THC are linked to cannabis use disorder and increased susceptibility to psychiatric illness. Patients with bipolar disorder who use marijuana also have a more adverse course of illness than those who do not use it.

