Low Frequency rTMS for Treatment-Resistant Auditory Hallucinations in Schizophrenia
Highlights from Posters Presented at the Society of Biological Psychiatry Meeting, April 27-29, 2023 in San Diego:
J. Brunelin gave patients “30 sessions of 1 Hz rTMS (repetitive Transcranial magnetic stimulation) over 3 weeks (360 pulses per sessions delivered with 60 sec ‘on’ and 30 sec ‘off’ at 110% of the resting motor threshold, 2 sessions per day). Stimulations were applied over the left temporoparietal junction.” Severity of auditory verbal hallucinations decreased (p=0.003) including in the 9 patients included with clozapine-resistant symptoms.
Good Outcomes Among Patients with Major Depressive Disorder Treated with Transcranial Magnetic Stimulation in a Large Study
At the 2021 meeting of the Society of Biological Psychiatry (SOBP), researcher Harold Sackeim and colleagues reported on data collected from patients in clinical treatment for major depression who received transcranial magnetic stimulation (TMS) at 103 practice sites. A total of 5,010 depressed patients were included in the intent-to-treat sample, and 3,814 completed the study, meaning that they either reached remission or were treated at least 20 times and went through a final assessment. “Response (58–83%) and remission (28–62%) rates were notably high across self-report and clinician-administered assessments,” and women had better outcomes than men. Sackeim and colleagues concluded, “Strong efficacy and the low side effect and medical risk profile suggest that TMS be evaluated as a first-line treatment for [major depressive disorder].”
Accelerated Intermittent Theta-Burst Stimulation (aiTBS) Quickly Improved Treatment-Resistant Depression
At the 2021 meeting of the Society of Biological Psychiatry (SOBP), researcher Nolan Williams and colleagues described a sham-controlled trial of accelerated intermittent theta-burst stimulation (aiTBS) in patients with treatment-resistant depression. Theta burst stimulation is a specific protocol for rTMS, or repeated transcranial magnetic stimulation, a non-invasive form of brain stimulation. Magnets placed on a patient’s head provide bursts of high-frequency stimulation to the brain.
Participants in the study received 50 sessions over 5 days of either aiTBS or a sham procedure. Among those who received the real aiTBS treatment, 85.7% saw an improvement in their treatment-resistant depression, compared to only 26.7% of those in the sham group. AiTBS produced a rapid antidepressant response, which Williams and colleagues suggest could be useful for the treatment of patients in emergency rooms or inpatient settings.
AiTBS Superior to ECT in Small Study
Researchers Erica Jensen and Nolan Williams reported in abstracts of a paper that they were to present at the 2020 meeting of the Society of Biological Psychiatry in May that daily sessions of accelerated intermittent theta burst transcranial stimulation (aiTBS) over five or more days produced better results in 15 patients hospitalized for depression and suicidality than in matched patients who received electro-convulsive therapy.
AiTBS is a form of repeated transcranial magnetic stimulation (rTMS), in which a magnetic coil is applied to a patient’s scalp, producing electrical changes in the brain.
The aiTBS treatment was delivered to the left dorsolateral prefrontal cortex. It consisted of 1800 pulses per session, at 80% of a patient’s resting motor threshold with a 50-minute inter-session interval.
The patients in the study were matched (for age, gender, and treatment resistance) to patients who were hospitalized and given ECT. Among patients who received aiTBS and were discharged after an average of 8.4 days, there was an 86% response rate and a 73% remission rate. Among the patients who received ECT, who were discharged after an average of 22.3 days, there was a 53% response rate and a 40% remission rate. With further ECT, response and remission rates increased to 73% and 67%. Time to remission was 3.5 days with aiTBS and 31.3 days for ECT. The investigators concluded conservatively, “Our results suggest that aiTBS could have comparable efficacy to ECT, with potentially faster resolution of acute severe depression.”
Editor’s Note: ECT has been the gold standard treatment for severe depression and suicidality and now we may have a platinum comparator. If these findings are replicated, they could represent a paradigm shift in the treatment of severe depression. Hopefully, this novel form of rTMS will be fast-tracked for approval by the Food and Drug Administration (FDA).
Three-Minute ‘Theta Burst’ Treatment as Effective as 37-Minute RTMS
A variation on repeated transcranial magnetic stimulation (rTMS) called intermittent theta burst stimulation (iTBS) may be able to deliver the same benefits in a tenth of the time. RTMS is a non-invasive treatment in which a magnetic coil placed near the skull transmits electrical signals to the brain. It is effective in depression and has been shown to improve aspects of schizophrenia, autism, and addictions as well.
A typical rTMS session lasts for 37.5 minutes and consists of high frequency (10 Hz) stimulation. Access to the treatment remains somewhat limited, so the newer form of iTBS treatment may help more people access treatment by allowing clinicians to treat more patients in a day.
The 2018 study, published by Daniel Blumberger and colleagues in the journal The Lancet, compared iTBS to standard rTMS and evaluated the effectiveness, safety, and tolerability of the new treatment compared to the old. 414 patients aged 18–65 with major depression that had persisted despite treatment with several antidepressant options were randomized to receive either iTBS or rTMS delivered to their left dorsolateral prefrontal cortex. They received the given treatment five days/week for four to six weeks.
Patients who received iTBS showed a nearly identical level of improvement in depression to those who received rTMS. Self-reports of pain intensity were worse among those who received iTBS, but the dropout rate was not higher for that group. Headaches were the most common side effect reported, and rates were similar across both groups. The authors judged iTBS to be a comparable, non-inferior alternative to rTMS for people with major depression.
Among participants who received iTBS, depression improved significantly, with 32 percent reporting a remission of depression symptoms. Those who received standard rTMS had a remission rate of 27 percent.
Medical Device May Treat Alzheimer’s Disease
A recently completed clinical trial suggests that NeuroAD, a treatment system that combines transcranial magnetic stimulation and cognitive training targeted at brain regions affected by Alzheimer’s disease, may be effective at treating mild to moderate cases of the illness.
Neuronix Ltd, the company that produces the device used to deliver transcranial magnetic stimulation in the trial, plans to seek Food and Drug Administration approval for NeuroAD. It would be the first device approved for the treatment of Alzheimer’s in the US. The device is already in use in Europe and Asia.
In the clinical trial, 131 patients received six weeks of the NeuroAD treatment or a sham treatment used as a comparison. Those participants who received the real intervention performed better on an assessment of Alzheimer’s and experienced minimal side effects.
In transcranial magnetic stimulation, a non-invasive procedure, magnets placed near the skull stimulate electrical impulses in the brain. This activates neurons, releasing excitatory transmitters and brain-derived neurotrophic factor (BDNF), which is important for new synapse formation and long-term learning and memory.
Editor’s Note: This editor (Robert Post) has long advocated the use of repeated transcranial magnetic stimulation (rTMS) with simultaneous cognitive behavioral or other positive therapy to activate and enhance specific neural circuits and relieve depression. The trial of NeuroAD adds evidence of the positive effects of this approach in domains other than depression. Cognitive training enhanced by rTMS may be helpful with a variety of cognitive difficulties.
RTMS May Treat Cocaine Addictions
 In a pilot study, repeated transcranial magnetic stimulation (rTMS) reduced cocaine cravings and usage among people with cocaine addiction.
In a pilot study, repeated transcranial magnetic stimulation (rTMS) reduced cocaine cravings and usage among people with cocaine addiction.
RTMS is a non-invasive treatment in which a magnetic coil placed near the skull transmits electrical signals to the brain. It is an effective treatment for depression, and there is growing evidence that it may also be able to treat addictions.
Participants in the pilot study by researcher Antonello Bonci and colleagues received rTMS directed at their dorsolateral prefrontal cortex or pharmacological treatments (including medications to manage depression, anxiety, and sleep problems) over a 29-day study period. Among the rTMS recipients, 69% remained cocaine-free during the study period, compared to only 19% of those treated with medications. Those who received rTMS also reported fewer cravings.
There were few side effects among those who received rTMS, and there was a 100% compliance rate among the 32 participants, meaning they all showed up for each of their sessions.
Bonci and colleagues are working on a larger study that will compare rTMS treatment to a sham procedure rather than to a medication regime.
New Study Suggests RTMS Can Reduce Cocaine Use and Cocaine Cravings
Repeated transcranial magnetic stimulation, or rTMS, is a non-invasive treatment in which a magnetic coil placed near the skull transmits electrical signals to the brain. It is an effective treatment for depression, and now it appears it may also be useful in the treatment of addictions.
A pilot study by Alberto Terraneo and colleagues published in European Neuropsychopharmacology in 2016 compared rTMS treatment delivered to the dorsolateral prefrontal cortex to pharmacological treatment in 32 patients who wanted to stop using cocaine. Those in the rTMS group received one session of the treatment per day for five days, followed by one session per week for three weeks. Those who received rTMS had a higher number of cocaine-free urine tests than those who had been treated with pharmacological treatments. Among those who received rTMS, 69% had a positive outcome, compared to 19% of the control group. RTMS also reduced cravings for cocaine. Both treatments improved depression.
Antonello Bonci, another author of the study who is also scientific director at the US National Institute on Drug Abuse, suggested that rTMS may work by “scrambling” the pattern of neural activity that leads to cocaine craving.
Now that there is some evidence suggesting that rTMS may be useful in the treatment of addictions, the researchers are planning a placebo-controlled study of rTMS treatment for cocaine use, in which they will give some patients a sham treatment instead of real rTMS.
Other studies are examining whether rTMS can be used to treat smoking and alcohol use disorders in addition to depression.
RTMS Improves Executive Function in Kids with Schizophrenia and Autism
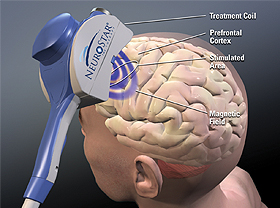 Researcher Stephanie Ameis reported at the 2015 meeting of the American Academy of Child and Adolescent Psychiatry that following repeated transcranial magnetic stimulation (rTMS), a treatment in which a magnetic coil placed over the scalp delivers electric pulses to the brain, children with schizophrenia and autism spectrum disorders showed improvements in executive function, including working memory. The rTMS treatment targeted the left dorsolateral prefrontal cortex.
Researcher Stephanie Ameis reported at the 2015 meeting of the American Academy of Child and Adolescent Psychiatry that following repeated transcranial magnetic stimulation (rTMS), a treatment in which a magnetic coil placed over the scalp delivers electric pulses to the brain, children with schizophrenia and autism spectrum disorders showed improvements in executive function, including working memory. The rTMS treatment targeted the left dorsolateral prefrontal cortex.
RTMS for Depression Increases Volume of Specific Brain Regions
Repeated transcranial magnetic stimulation (rTMS) is a treatment for depression in which magnets placed near the skull stimulate electrical impulses in the brain. In a poster presented at the 2015 meeting of the Society of Biological Psychiatry, Martin Lan and colleagues presented results of the first study of structural changes in the brain following rTMS.
In the study, 27 patients in an episode of major depression underwent magnetic resonance brain scans before and after receiving rTMS treatment over their left prefrontal cortices. Lan and colleagues reported that several cortical regions related to cognitive appraisal, the subjective experience of emotion, and self-referential processing increased in volume following rTMS treatment: the anterior cingulate, the cingulate body, the precuneous, right insula, and gray matter in the medial frontal gyrus. The increases ranged from 5.3% to 15.7%, and no regions decreased in volume. More than 92% of the participants showed increased gray matter in all of these regions.
The brain changes were not correlated with antidepressant response to rTMS, but suggest a possible mechanism by which rTMS is effective in some people. Lan and colleagues concluded that rTMS likely had neuroplastic effects in areas of the brain that are important for emotion regulation.