Accelerated Intermittent Theta-Burst Stimulation (aiTBS) Quickly Improved Treatment-Resistant Depression
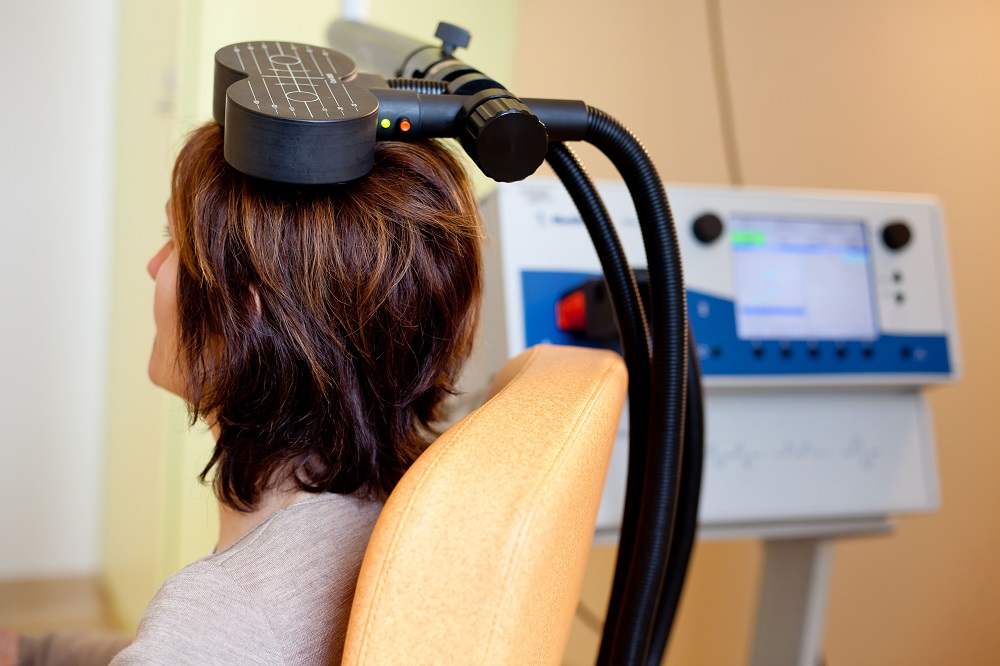
At the 2021 meeting of the Society of Biological Psychiatry (SOBP), researcher Nolan Williams and colleagues described a sham-controlled trial of accelerated intermittent theta-burst stimulation (aiTBS) in patients with treatment-resistant depression. Theta burst stimulation is a specific protocol for rTMS, or repeated transcranial magnetic stimulation, a non-invasive form of brain stimulation. Magnets placed on a patient’s head provide bursts of high-frequency stimulation to the brain.
Participants in the study received 50 sessions over 5 days of either aiTBS or a sham procedure. Among those who received the real aiTBS treatment, 85.7% saw an improvement in their treatment-resistant depression, compared to only 26.7% of those in the sham group. AiTBS produced a rapid antidepressant response, which Williams and colleagues suggest could be useful for the treatment of patients in emergency rooms or inpatient settings.
Scientific Mechanisms of Rapid-Acting Antidepressants
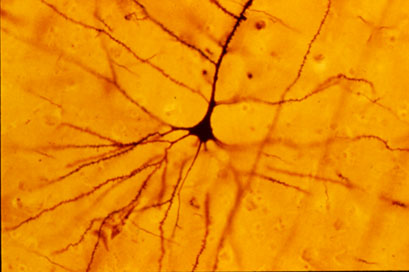
A pyramidal cell (Photo by Bob Jacobs, Laboratory of Quantitative Neuromorphology Department of Psychology Colorado College)
At a recent symposium, researcher Francis McMahon provided electrophysiological evidence that several different types of rapid-acting antidepressants—low-dose ketamine, scopolamine, and rapastinel (a partial agonist of the neurotransmitter NMDA)—act by decreasing the inhibitory effects of GABAergic interneurons on excitatory neurons called pyramidal cells, thus increasing synaptic firing.
Researcher Ronald Duman further dissected these effects, showing that ketamine and its active metabolite norketamine reduce the steady firing rate of GABA interneurons by blocking NMDA receptors, while the partial agonist rapastinel acts on the glutamate neurons directly, and both increase the effects of a type of glutamate receptors known as AMPA. These effects were demonstrated using a virus to selectively knock out GluN2B glutamate receptor subunits in either GABA interneurons or glutamate neurons.
Increasing AMPA activity increases synapse number and function and also increases network connectivity, which can reverse the effects of stress. Duman and colleagues further showed that when light is used to modulate pyramidal cells (a process called optogenetic stimulation) in the medial prefrontal cortex, different effects could be produced. Stimulating medial prefrontal cortex cells that contained dopamine D1 receptors, but not D2 receptors, produced rapid and sustained antidepressant effects. Conversely, inhibiting these neurons blocked the antidepressant effects of ketamine. Stimulating the terminals of these D1-containing neurons in the basolateral nucleus of the amygdala was sufficient to reproduce the antidepressant effects. These data suggest that stimulation of glutamate D1 pyramidal neurons from the medial prefrontal cortex to the basolateral nucleus of the amygdala is both necessary and sufficient to produce the antidepressant effects seen with ketamine treatment.
Researcher Hailan Hu reported that NMDA glutamate receptors drive the burst firing of lateral habenula (LHb) neurons, which make up the depressogenic or “anti-reward center” of the brain and appear to mediate anhedonic behavior (loss of interest or enjoyment) in animal models of depression. Ketamine blocks the burst firing of the LHb neurons, which disinhibits monoamine reward centers, enabling ketamine’s rapid-onset antidepressant effects. This may occur because inhibitory metabotropic glutamate receptors (mGluR-2) are activated, decreasing the release of glutamate.
MGluR-2 may also help explain the antidepressant effects of acetyl-L-carnitine supplements. L-carnitine is an amino acid that is low in the blood of depressed patients. The supplement acetyl-L-carnitine (ACL) activates the DNA promoter for mGluR-2, increasing its production and thus decreasing excess glutamate release. The acetyl group of the ACL binds to the DNA promoter for mGluR-2, thus this process seems to be epigenetic. Epigenetic mechanisms affect the structure of DNA and can be passed on to offspring even though they are not encoded in the DNA’s genetic sequence.
Mixed Findings for Intranasal Ketamine
The drug ketamine can rapidly and temporarily improve depression when delivered intravenously. Researchers have been working on extending ketamine’s effects and finding easier ways of delivering the medication. One new delivery method under investigation is nasal spray, which could be used repeatedly to extend ketamine’s effects.
Unfortunately, researcher Colleen Loo reported in the Journal of Psychopharmacology in 2018 that a pilot study of self-administered intranasal ketamine for severe depression was suspended when 5 of the 10 participants had side effects that included high blood pressure, psychotic symptoms, and motor incoordination that made them unable to keep using the spray. Early in the four-week study, dosage was adjusted to leave more time between sprays, but this was not enough to prevent the problems with side effects.
Loo said that the nasal spray version of ketamine has complications including variations in absorption among different people and on different days, depending on factors like mucus in the nose and exact application techniques. Its rapid absorption into the bloodstream could lead to high peak levels in certain people.
Loo and colleagues had previously found that elderly patients receiving injections of ketamine under the skin required highly individualized dosing to avoid side effects. This may also be the case with nasal spray.
While Loo’s study found intranasal ketamine infeasible for the moment, Janssen Research and Development, a pharmaceutical company owned by Johnson & Johnson, reported positive results in phase 3 clinical trials of intranasal esketamine (a component of ketamine) at the annual meeting of the American Psychiatric Association in May. Researchers for Janssen reported that intranasal esketamine was highly effective for depression and well-tolerated both in acute treatment and over a year-long period. Janssen is now pursing approval for the drug from the US Food and Drug Administration.
An Inflammatory State Impedes Treatment for Bipolar Disorder
 A 2017 study by in the Journal of Clinical Psychiatry links inflammation to a poor antidepressant response in bipolar disorder. Many previous studies have found that elevated inflammatory markers are common in mood disorders, and that an inflammatory state seems to prevent response to certain therapies.
A 2017 study by in the Journal of Clinical Psychiatry links inflammation to a poor antidepressant response in bipolar disorder. Many previous studies have found that elevated inflammatory markers are common in mood disorders, and that an inflammatory state seems to prevent response to certain therapies.
Researcher Francesco Benedetti and colleagues report that high levels of inflammatory cytokines (a type of small proteins) predicted a worse response to treatment with sleep deprivation and light therapy for bipolar depression. This treatment typically brings about a rapid antidepressant response.
Benedetti and colleagues measured 15 immune-regulating compounds in 37 patients who were experiencing an episode of bipolar depression and 24 healthy volunteers. Among those participants with bipolar disorder, 84% had a history of non-response to medication. Twenty-three of the 37 patients, or 62%, responded to the sleep deprivation/light therapy combination. Those who did not had higher levels of five cytokines: interleukin-8, monocyte chemoattractant protein-1, interferon-gamma, interleukin-6, and tumor necrosis factor-alpha.
Body mass index was correlated with cytokine levels and also reduced response to the treatment.
The finding supports a link between the immune system and mood disorders. Evaluating a patient’s level of inflammation may, in the future, allow doctors to predict the patient’s response to a given therapy. Patients with high levels of inflammation might benefit most from treatments that target their immune system.
Bloomberg Covers Ketamine as a Depression Treatment
Over the years, we’ve tried to keep readers updated about ketamine‘s use as a fast-acting antidepressant. Bloomberg Business recently covered how the drug is being used by some to treat depression.
Laughing Gas Reduces Depression for Up to 24 Hours
In a proof-of-concept study presented at the 2015 meeting of the Society of Biological Psychiatry, Charles R. Conway and Peter Nagele showed that an hour of 50% oxygen/50% nitrous oxide reduced depression more than placebo as measured 2 hours and 24 hours later. Twenty patients were randomized to receive the laughing gas combination or a placebo combination made up of 50% oxygen/50% nitrogen. In the laughing gas group, four patients responded to the treatment and three patients achieved remission, compared to only one patient responding in the placebo group.
Like the anesthetic ketamine, which can bring about rapid but temporary antidepressant effects when delivered intravenously, nitrous oxide is an NMDA receptor antagonist.
Rapid-Onset Antidepressant Treatments
At the International College of Neuropsychopharmacology (CINP) World Congress of Neuropsychopharmacology in 2014, several presentations and posters discussed treatments that bring about rapid-onset antidepressant effects, including ketamine, isoflurane, sleep deprivation, and scopolamine.
Ketamine’s Effects
Multiple studies, now including more than 23 according to researcher William “Biff” Bunney, continue to show the rapid-onset antidepressant efficacy of intravenous ketamine, usually at doses of 0.5 mg/kg over 40 minutes. Response rates are usually in the range of 50–70%, and effects are seen within two hours and last several days to one week. Even more remarkable are the six studies (two double-blind) reporting rapid onset of antisuicidal effects, often within 40 minutes and lasting a week or more. These have used the same doses or lower doses of 0.1 to 0.2mg/kg over a shorter time period.
Attempts to sustain the initial antidepressant effects include repeated ketamine infusions every other day up to a total of six infusions, a regimen in which typically there is no loss of effectiveness. Researcher Ronald Duman is running a trial of co-treatment with ketamine and lithium, since both drugs block the effects of GSK-3, a kinase enzyme that regulates an array of cellular functions, and in animals the two drugs show additive antidepressant effects. In addition, lithium has been shown to extend the acute antidepressant effects of one night of sleep deprivation, which are otherwise reversed by a night of recovery sleep.
Ketamine’s effects are related to the neurotransmitter glutamate, for which there are several types of receptors, including NMDA and AMPA. Ketamine causes a large burst of glutamate presumably because it blocks NMDA glutamate receptors on inhibitory interneurons that use the neurotransmitter GABA, causing glutamatergic cells to lose their inhibitory input and fire faster. While ketamine blocks the effects of this glutamate release at NMDA receptors, actions at AMPA receptors are not blocked, and AMPA activity actually increases. This increases brain-derived neurotrophic factor (BDNF), which is also required for the antidepressant effects of ketamine. Ketamine also increases the effects of mTOR, a kinase enzyme that regulates cell growth and survival, and if these are blocked with the antibiotic rapamycin, antidepressant effects do not occur.
In animal studies, ketamine increases dendritic spine growth and rapidly reverses the effects of chronic mild unpredictable stressors on the spines (restoring their mature mushroom shape and increasing their numbers), effects that occur within two hours in association with its rapid effects on behaviors that resemble human depression.
About 50–70% of treatment-resistant depressed patients respond to ketamine. However, about one-third of the population has a common genetic variation of BDNF in which one or both valine amino acids that make up the typical val-66-val allele are replaced with methionine (producing val-66-met proBDNF or met-66-met proBDNF). The methionine variations result in the BDNF being transported less easily within the cell. Patients with these poorly functioning alleles of BDNF are less likely to get good antidepressant effects from treatment with ketamine.
Ketamine in Animal Studies
Researcher Pierre Blier reviewed the effects of ketamine on the neurotransmitters serotonin, norepinephrine, and dopamine. In rodents, a swim stress test is used to measure depression-like behavior. Researchers record how quickly the rodents give up trying to get out of water and begin to float instead. Blier found that ketamine’s effects on swim stress were dependent on all three neurotransmitters. For dopamine, ketamine’s effects were dependent on increases in the number of dopamine cells firing, not on the firing rate, and for norepinephrine, ketamine’s effects were dependent on increases in burst firing patterns. Each of these effects was dependent on glutamate activity at AMPA receptors. Given these effects, Blier believes that using ketamine as an adjunct to conventional antidepressants that tend to increase these neurotransmitters may add to its clinical effectiveness.
Important Anecdotal Clinical Notes
Blier reported having given about 300 ketamine infusions to 25 patients, finding that two-thirds of these patients responded, including one-third who recovered completely, while one-third did not respond to the treatment. Patients received an average of 12 infusions, not on a set schedule, but according to when they began to lose response to the last ketamine infusion. If a patient had only a partial response, Blier gave the next ketamine treatment at a faster rate of infusion and was able to achieve a better response. These clinical observations are among the first to show that more than six ketamine infusions may be effective and well tolerated. Read more




