Pilot Study of Pramipexole for Anhedonia Symptoms in Unipolar Depression

At a recent scientific meeting, researcher Laura Hack described an open-label pilot study of pramipexole in participants with major depression including anhedonia, or inability to feel pleasure. Five participants with prominent anhedonia and major depression completed eight weeks of treatment with pramipexole, which activates dopamine D2 and D3 receptors. Four of the five who completed the study saw notable improvements in their anhedonia and quality of life.
The starting daily dose was 0.26 mg, which was increased every three days to a target dose of 2.0 mg per day. Two additional patients dropped out due to side effects (nausea, poor sleep, and headache).
Low activity in the ventral striatum, a part of the brain associated with decision-making and implicated in the brain’s reward system, correlated with the severity of the patients’ anhedonia. These results suggest that further studies are warranted.
Pramipexole has also shown positive effects in two small studies in bipolar depression. The drug is currently approved by the US Food and Drug Administration for the treatment of Parkinson’s disease and also works in restless legs syndrome.
New Type of Antipsychotic Drug for Schizophrenia Looks Promising
In a 2020 article in the New England Journal of Medicine, researcher Kenneth S. Koblan and colleagues described a new type of antipsychotic drug treatment for schizophrenia. Almost all other antipsychotic drugs block dopamine D2 receptors, while atypical antipsychotics also block the serotonin 5HT2 receptor. They are described as antagonists at these receptors.
In contrast, the new drug is an agonist or activator of two different receptors. The drug SEP-363856 (also called SEP-856) activates the trace amine–associated receptor 1 (TAAR1) and 5-hydroxytryptamine (or serotonin) type 1A (5-HT1A) receptors.
Blocking D2 receptors can cause Parkinson’s-like symptoms (such as tremor, masked faces, and impaired movement or speech) and other extrapyramidal side effects (such as slurred speech, slow movements, or restless legs.) In contrast, SEP-856 seems to have a better side effects profile than these types of drugs while also being highly effective.
Patients with an acute exacerbation of schizophrenia were assigned to receive either placebo or once-daily treatment with SEP-856 (either 50mg or 75mg) for four weeks. A total of 120 patients received SEP-856 while 125 received placebo.
Compared to the placebo group, the SEP-856 group showed significantly greater reductions on a scale of positive and negative symptoms of schizophrenia by the end of the four weeks. Side effects included some sleepiness and gastrointestinal symptoms, but the incidence of extrapyramidal symptoms and changes in the levels of lipids, glycated hemoglobin, and prolactin were similar in both groups. There was one sudden death from cardiac causes in the SEP-856 group, which was not thought to be drug-related.
Editor’s Note: This drug acting on trace amine–associated receptor 1 (TAAR1) and 5HT1A receptors could herald a new and better tolerated type of antipsychotic. It is also being studied for psychosis in Parkinson’s disease. Since all of the antipsychotics that treat schizophrenia have also shown antimanic efficacy, we look forward to future studies of this unique drug in patients with mania.
Scientific Mechanisms of Rapid-Acting Antidepressants
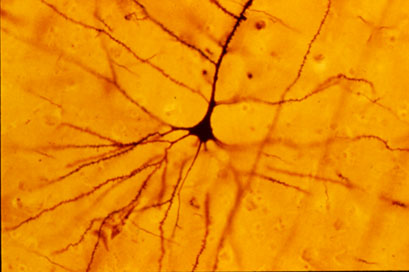
A pyramidal cell (Photo by Bob Jacobs, Laboratory of Quantitative Neuromorphology Department of Psychology Colorado College)
At a recent symposium, researcher Francis McMahon provided electrophysiological evidence that several different types of rapid-acting antidepressants—low-dose ketamine, scopolamine, and rapastinel (a partial agonist of the neurotransmitter NMDA)—act by decreasing the inhibitory effects of GABAergic interneurons on excitatory neurons called pyramidal cells, thus increasing synaptic firing.
Researcher Ronald Duman further dissected these effects, showing that ketamine and its active metabolite norketamine reduce the steady firing rate of GABA interneurons by blocking NMDA receptors, while the partial agonist rapastinel acts on the glutamate neurons directly, and both increase the effects of a type of glutamate receptors known as AMPA. These effects were demonstrated using a virus to selectively knock out GluN2B glutamate receptor subunits in either GABA interneurons or glutamate neurons.
Increasing AMPA activity increases synapse number and function and also increases network connectivity, which can reverse the effects of stress. Duman and colleagues further showed that when light is used to modulate pyramidal cells (a process called optogenetic stimulation) in the medial prefrontal cortex, different effects could be produced. Stimulating medial prefrontal cortex cells that contained dopamine D1 receptors, but not D2 receptors, produced rapid and sustained antidepressant effects. Conversely, inhibiting these neurons blocked the antidepressant effects of ketamine. Stimulating the terminals of these D1-containing neurons in the basolateral nucleus of the amygdala was sufficient to reproduce the antidepressant effects. These data suggest that stimulation of glutamate D1 pyramidal neurons from the medial prefrontal cortex to the basolateral nucleus of the amygdala is both necessary and sufficient to produce the antidepressant effects seen with ketamine treatment.
Researcher Hailan Hu reported that NMDA glutamate receptors drive the burst firing of lateral habenula (LHb) neurons, which make up the depressogenic or “anti-reward center” of the brain and appear to mediate anhedonic behavior (loss of interest or enjoyment) in animal models of depression. Ketamine blocks the burst firing of the LHb neurons, which disinhibits monoamine reward centers, enabling ketamine’s rapid-onset antidepressant effects. This may occur because inhibitory metabotropic glutamate receptors (mGluR-2) are activated, decreasing the release of glutamate.
MGluR-2 may also help explain the antidepressant effects of acetyl-L-carnitine supplements. L-carnitine is an amino acid that is low in the blood of depressed patients. The supplement acetyl-L-carnitine (ACL) activates the DNA promoter for mGluR-2, increasing its production and thus decreasing excess glutamate release. The acetyl group of the ACL binds to the DNA promoter for mGluR-2, thus this process seems to be epigenetic. Epigenetic mechanisms affect the structure of DNA and can be passed on to offspring even though they are not encoded in the DNA’s genetic sequence.
Schizophrenia: The Importance of Catching It Early
By the time psychosis appears in someone with schizophrenia, biological changes associated with the illness may have already been present for years. A 2015 article by R.S. Kahn and I.E. Sommer in the journal Molecular Psychiatry describes some of these abnormalities and how treatments might better target them.
One such change is in brain volume. At the time of diagnosis, schizophrenia patients have a lower intracranial volume on average than healthy people. Brain growth stops around age 13, suggesting that reduced brain growth in people with schizophrenia occurs before that age.
At diagnosis, patients with schizophrenia show decrements in both white and grey matter in the brain. Grey matter volume tends to decrease further in these patients over time, while white matter volume remains stable or can even increase.
Overproduction of dopamine in the striatum is another abnormality seen in the brains of schizophrenia patients at the time of diagnosis.
Possibly years before the dopamine abnormalities are observed, underfunctioning of the NMDA receptor and low-grade brain inflammation occur. These may be linked to cognitive impairment and negative symptoms of schizophrenia such as social withdrawal or apathy, suggesting that there is an at-risk period before psychosis appears when these symptoms can be identified and addressed. Psychosocial treatments such as individual, group, or family psychotherapy and omega-3 fatty acid supplementation have both been shown to decrease the rate of conversion from early symptoms to full-blown psychosis.
Using antipsychotic drugs to treat the dopamine abnormalities is generally successful in patients in their first episode of schizophrenia. Use of atypical antipsychotics is associated with less brain volume loss than use of the older typical antipsychotics. Treatments to correct the NMDA receptor abnormalities and brain inflammation, however, are only modestly effective. (Though there are data to support the effectiveness of the antioxidant n-acetylcysteine (NAC) on negative symptoms compared to placebo.) Kahn and Sommer suggest that applying treatments when cognitive and social function begin to be impaired (rather than waiting until psychosis appears) could make them more effective.
The authors also suggest that more postmortem brain analyses, neuroimaging studies, animal studies, and studies of treatments’ effects on brain abnormalities are all needed to clarify the causes of the early brain changes that occur in schizophrenia and identify ways of treating and preventing them.
Cariprazine More Potent Than Aripiprazole at Dopamine D3 Receptors
Cariprazine is a new atypical antipsychotic that has positive results in the treatment of schizophrenia, bipolar mania and depression, and as an add-on treatment to antidepressants in unipolar depression. In a recent study by Frank Tarazi and colleagues, cariprazine shared some actions with aripiprazole (trade name Abilify), which is approved by the Federal Drug Administration as an adjunctive treatment for unipolar depression in addition to treating mania in bipolar disorder and schizophrenia.
Both cariprazine and aripiprazole partially block dopamine receptors. They allow a little stimulation of the receptors (activating or agonist effects) while preventing dopamine from binding there. With ongoing treatment, this blocking action prompts an increase in the number of dopamine receptors to compensate for the blocking.
In rats, both drugs led to increases in several types of dopamine receptors, but cariprazine did so at lower doses. Following 28 days of treatment with aripiprazole at doses of 5–15 mg/kg, rats had higher levels of D2 and D4 receptors in several brain regions than other rats that received no drug. Higher doses of aripiprazole also increased D3 receptors in some regions, indicating that the drug works less well at those receptors. Lower doses of cariprazine (0.06–0.6 mg/kg) increased D2 and D4 receptors significantly, and increased D3 receptors more than aripiprazole did, and in a greater number of forebrain regions.
Among antipsychotic drugs, both aripiprazole and cariprazine are unique in providing a little stimulation (partial agonist effects) at dopamine receptors, while all others drugs in the class are pure blockers (antagonists) of dopamine receptors.
The researchers concluded that cariprazine was more potent than aripiprazole at dopamine receptors, especially D3 receptors.
Methamphetamine Kills Dopamine Neurons in the Midbrain of Mice
Epidemiological studies have linked methamphetamine use to risk of Parkinson’s disease, and animal studies of the illicit drug have shown that it harms dopamine neurons. A 2014 study by Sara Ares-Santos et al. in the journal Neuropsychopharmacology compared the effects of repeated low or medium doses to those of a single high dose on mice. Loss of dopaminergic terminals, where dopamine is released, was greatest after three injections of 10mg/kg given at three-hour intervals, followed by three injections of 5 mg/kg at three-hour intervals, and a one-time dose of 30mg/kg. All of the dosages produced similar rates of degeneration of dopamine neurons via necrosis (cell destruction) and apoptosis (cell suicide) in the substantia nigra pars compacta (the part of the brain that degenerates in Parkinson’s disease) and the striatum.
Methamphetamine Kills Dopamine Neurons in the Midbrain of Mice
Epidemiological studies have linked methamphetamine use to risk of Parkinson’s disease, and animal studies of the illicit drug have shown that it harms dopamine neurons. A 2014 study by Sara Ares-Santos et al. in the journal Neuropsychopharmacology compared the effects of repeated low or medium doses to those of a single high dose on mice. Loss of dopaminergic terminals, where dopamine is released, was greatest after three injections of 10mg/kg given at three-hour intervals, followed by three injections of 5 mg/kg at three-hour intervals, and a one-time dose of 30mg/kg. All of the dosages produced similar rates of degeneration of dopamine neurons via necrosis (cell destruction) and apoptosis (cell suicide) in the substantia nigra pars compacta (the part of the brain that degenerates in Parkinson’s disease) and the striata.
New Research on Ketamine
 The drug ketamine can produce antidepressant effects within hours when administered intravenously.
The drug ketamine can produce antidepressant effects within hours when administered intravenously.
Finding an Appropriate Control
Comparing ketamine to placebo has challenges because ketamine produces mild dissociative effects (such as a feeling of distance from reality) that are noticeable to patients. At the 2013 meeting of the Society of Biological Psychiatry, James W. Murrough and collaborators at the Mount Sinai School of Medicine reported their findings from the first controlled trial of intravenous ketamine in depression that uses an active control, the short-acting benzodiazepine midazolam, which has sedative effects and decreases anxiety, but is not known as an antidepressant. On virtually all measures intravenous ketamine was a more effective antidepressant following 2 infusions per week.
These data help dispel one of the criticisms of intravenous ketamine, that studies of the drug have not been sufficiently blinded (when patients and medical staff are kept from knowing which patients receive an active treatment and which are in the placebo control group) and that the lack of an appropriate active placebo contributed to the dramatic findings about ketamine’s antidepressant effects. It now appears that these criticisms have been appropriately answered and that intravenous ketamine is highly effective not only in comparison to placebo but also to an active comparator.
This research was presented as a poster at the meeting and published as abstract #442 in the meeting supplement to the journal Biological Psychiatry, Volume 73, Number 9S, and was also published in the Archives of General Psychiatry in 2013.
Slowing Down Ketamine Infusions to Reduce Side Effects
Ketamine is commonly given in 40-minute intravenous infusions. Timothy Lineberry from the Mayo Clinic reported in Abstract #313 from the meeting that slower infusions of ketamine over 100 minutes were also effective in producing antidepressant effects in patients with treatment-resistant depression. Lineberry’s research group used the slower infusion in order to increase safety and decrease side effects, such as the dissociative effects discussed above. In the 10 patients the group studied, they observed a response rate of 80% and a remission rate of 50% (similar to ketamine’s effects with 40-minute infusions).
Family or Personal History of Alcohol Dependence Predicts Positive Response to Ketamine in Depression
Mark J. Niciu and collaborators at the NIMH reported in Abstract #326 that a personal or family history of alcohol dependence predicted a positive response to IV ketamine in patients with unipolar depression.
Ketamine Acts on Monoamines in Addition to Glutamate
Ketamine’s primary action in the nervous system is to block glutamate NMDA receptors in the brain. In addition to its effects on glutamate, it may also affect the monoamines norepinephrine and dopamine. Kareem S. El Iskandarani et al. reported in Abstract #333 that in a study of rats, ketamine increased the firing rate of norepinephrine neurons in a part of the brain called the locus coeruleus and also increased the number of spontaneous firing dopamine cells in the ventral tegmental area of the brain.
Editor’s Note: These data showing that ketamine increased the activity of two monoamines could help explain ketamine’s ability to induce rapid onset of antidepressant effects, in addition to its ability to immediately increase brain-derived neurotrophic factor (BDNF, important for long-term memory and the creation of new synapses) and to restore healthy mushroom-shaped spines on the dendrites of neurons in the prefrontal cortex.
Moclobemide May Help Depression and Post-Partum Blues
Monoamine oxidase inhibitors (MAO-Is) are a type of antidepressant that is often effective for people with anxious depression or comorbid panic attacks, especially when other antidepressants don’t work. This may be because MAO-Is work on all three neurotransmitter systems implicated in depression: dopamine, norepinephrine, and serotonin.

In a recent study presented at the 65th Annual Scientific Convention of the Society of Biological Psychiatry, Julia Sacher et al. found that six weeks of therapeutic doses of the MAO-I moclobemide (at doses of 300 mg twice a day) significantly decreased monoamine oxidase A, as measured by PET scans in brain regions implicated in mood disorders. In a comparison of moclobemide, placebo, and the herbal preparation St. John’s Wort, only moclobemide had a significant effect. Read more
Schizophrenia v. Bipolar Disorder: Different Risk Factors
 Robin Murray gave a plenary presentation at the 65th Annual Scientific Convention of the Society of Biological Psychiatry this year, in which he indicated that the genetic risk for schizophrenia and other major mental disorders may be overestimated. He suggested that even in identical twins there are considerable differences in incidence of major psychiatric illnesses, and sharing an environment could further inflate the appearance of genetic risk.
Robin Murray gave a plenary presentation at the 65th Annual Scientific Convention of the Society of Biological Psychiatry this year, in which he indicated that the genetic risk for schizophrenia and other major mental disorders may be overestimated. He suggested that even in identical twins there are considerable differences in incidence of major psychiatric illnesses, and sharing an environment could further inflate the appearance of genetic risk.
Evidence of some genetic vulnerability factors, such as neuregulin, disbindin, DISC-1, zinc finger transcription factors, and neurexin, has been replicated. However, these genes appear to contribute only about 1% of the vulnerability to schizophrenia or bipolar illness. Copy number variations (CNVs, extra or missing copies of a gene, which may alter its activity) and gene micro-deletions (in which small bits of DNA are missing) have been found in about 5% of patients with schizophrenia, in some patients with autism and mental disabilities, but not in those with bipolar illness.
Murray emphasized the importance of psychosocial and neuromotor markers of neural development in determining risk of subsequent major psychiatric illness, rather than the relatively weak genetic effects. He cited the work of MacCabe (2009), who collected information from 907,000 individuals in Sweden. Their scholastic achievement at age 15?16 was rated, and hospitalizations for psychosis were recorded from age 17?31. Of the 315,000 followed for the long term, 493 developed schizophrenia and 208 developed bipolar disorder.
Predictors of cognitive and motor development in these two major psychiatric illnesses appeared to differ. In those who went on to develop schizophrenia, there was a slower rate of motor development, receptive language, and overall IQ in adolescence, while in those who went on to develop bipolar disorder, there was a faster rate of motor development, more language facility, and higher IQ in adolescence.